Eli Lilly's Foundayo Weight Loss Pill Sees Strong Initial Demand
Written by Emily J. Thompson, Senior Investment Analyst
Updated: 5 days ago
0mins
Should l Buy LLY?
Source: Newsfilter
- Positive Market Response: Eli Lilly's weight loss pill Foundayo has attracted over 20,000 users in its first month, indicating strong market demand, as CEO Dave Ricks noted that this figure reflects consumer acceptance of the new drug.
- FDA Approval: Foundayo received FDA approval earlier this month, becoming the second oral GLP-1 drug after Novo Nordisk's Wegovy, which opens up new market opportunities for the company.
- Daily User Growth: Ricks revealed that more than 1,000 people are starting Foundayo each day, and while brand building will take time, this growth rate indicates strong market interest in the drug.
- User Demographics: Over 80% of Foundayo users are new to GLP-1 medications, suggesting that Eli Lilly has significant expansion potential in the new user market, which is expected to drive future sales growth.
Trade with 70% Backtested Accuracy
Stop guessing "Should I Buy LLY?" and start using high-conviction signals backed by rigorous historical data.
Sign up today to access powerful investing tools and make smarter, data-driven decisions.
Analyst Views on LLY
Wall Street analysts forecast LLY stock price to rise
20 Analyst Rating
18 Buy
2 Hold
0 Sell
Strong Buy
Current: 967.930
Low
950.00
Averages
1192
High
1500
Current: 967.930
Low
950.00
Averages
1192
High
1500
About LLY
Eli Lilly and Company is a medicine company, which discovers, develops, manufactures, and market products in a single business segment called human pharmaceutical products. The Company manufacture and distribute its products through facilities in the United States, including Puerto Rico, and in Europe and Asia. The Company’s products are sold in approximately 90 countries. Its Cardiometabolic Health products Basaglar; Humalog, Humalog Mix 75/25, Humalog U-100, Humalog U-200, Humalog Mix 50/50, insulin lispro, and others; Humulin, Humulin 70/30, and others; Jardiance; Mounjaro; Trulicity; Zepbound, and others. Its oncology products include Cyramza, Erbitux, Tyvyt, Verzenio, Retevmo, Jaypirca, and others. Its immunology products include Ebglyss, Olumiant, Omvoh, and Taltz. Its neuroscience products include Emgality and Kisunla. Its LillyDirect, a direct-to-patient digital health care platform, provides delivery of select Lilly medicines dispensed by third-party pharmacies to patients.
About the author

Emily J. Thompson
Emily J. Thompson, a Chartered Financial Analyst (CFA) with 12 years in investment research, graduated with honors from the Wharton School. Specializing in industrial and technology stocks, she provides in-depth analysis for Intellectia’s earnings and market brief reports.
- Revenue Surge: Eli Lilly's Q1 revenue soared 56% year-over-year to $19.8 billion, primarily driven by its blockbuster GLP-1 weight loss drugs, demonstrating robust market demand despite a 13% price decline offset by a 65% volume increase.
- Net Income Spike: Adjusted net income skyrocketed 155% to $7.7 billion, translating to $8.55 per share, significantly surpassing Wall Street's estimate of $6.79, reflecting the company's strong performance in the weight loss drug sector.
- Sales Growth Drivers: Sales of GLP-1 drugs Mounjaro and Zepbound surged 125% and 80%, reaching $8.7 billion and $4.2 billion respectively, becoming key growth drivers that further solidify the company's market leadership.
- Optimistic Outlook: Eli Lilly raised its 2023 revenue and adjusted EPS guidance to $82 billion to $85 billion and $35.50 to $37, indicating confidence in future growth, particularly with the newly launched GLP-1 pill Foundayo expected to expand market share.
See More
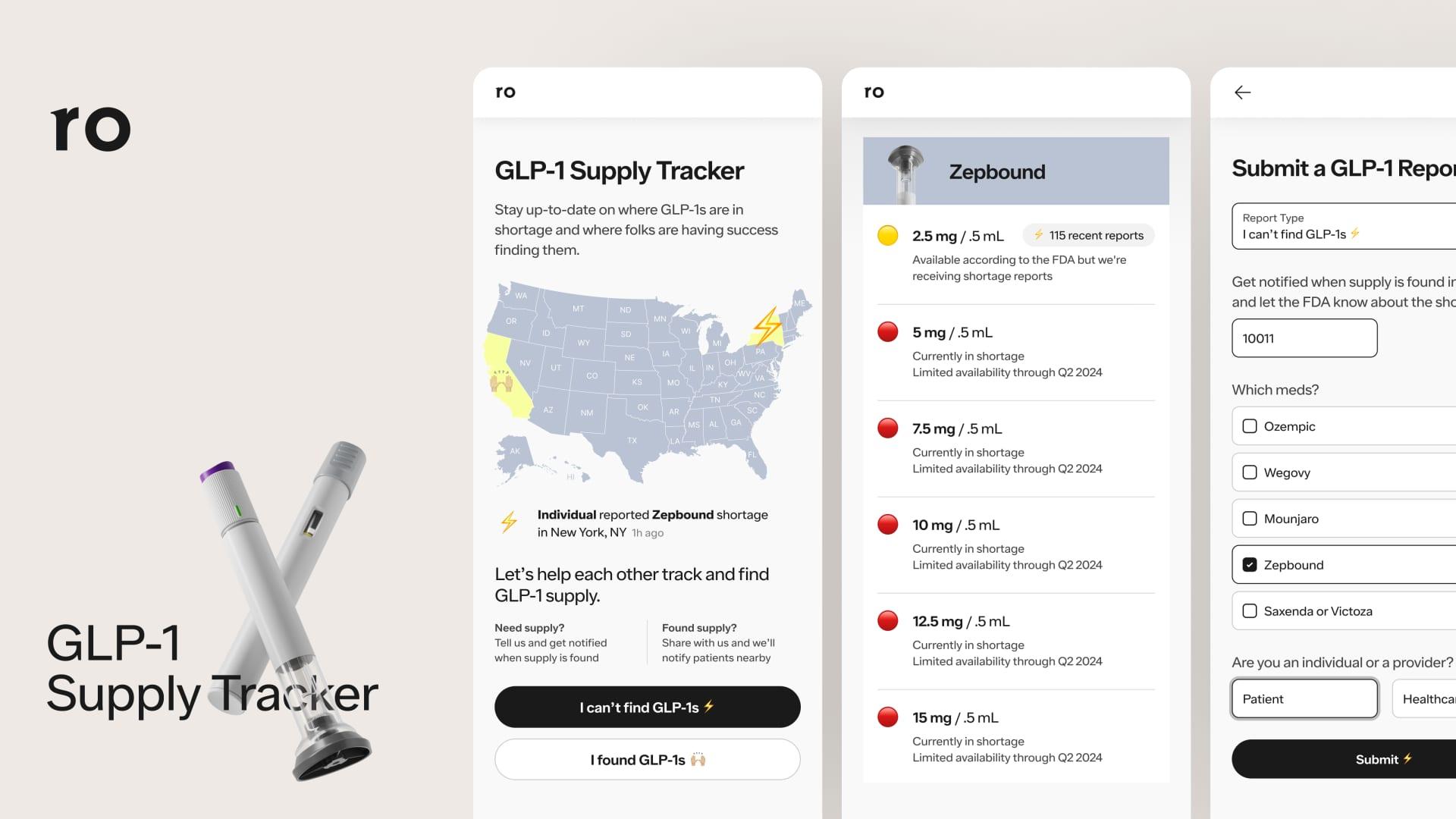
Health Canada Authorization: Health Canada has authorized the use of Lilly's drug, known as Skysunla (donanemab), for the treatment of early Alzheimer's disease.
New Treatment Option: This approval introduces a new therapeutic option aimed at addressing the needs of patients diagnosed with early-stage Alzheimer's.
See More
- DuPont Earnings Beat: DuPont reported better-than-expected earnings, with strong performance in healthcare, aerospace, and automotive sectors, although disruptions in the Middle East affected its water market; the organic sales guidance for the year was raised to 4%, indicating resilience against cost pressures, leading to a 2% increase in shares this morning.
- Eaton's Mixed Results: Eaton posted earnings and revenue beats, but shares fell approximately 4.5% due to conservative guidance and a miss in Electrical Americas; however, accelerating sales and order growth, along with backlog increases, suggest underlying strength, making the upcoming earnings call crucial for investor sentiment.
- Amazon Supply Chain Expansion: Amazon announced it would open its supply chain network to other companies, attracting major clients like Procter & Gamble, American Eagle, and 3M, which underscores its ambitions across retail, logistics, and health sectors, further solidifying its market position.
- Norwegian Cruise Line Guidance Cut: Norwegian Cruise Line issued a larger-than-expected guidance cut due to soft pricing, attributing below-optimal bookings to execution issues exacerbated by the Middle East conflict, prompting Goldman Sachs to lower its price target from $18 to $14, reflecting market concerns about its future performance.
See More
- Increased Market Competition: Amazon's announcement of a new initiative allowing external businesses to utilize its supply chain for deliveries led to significant stock declines for UPS and FedEx, dropping over 10% and 9% respectively, indicating investor concerns about intensified competition in the logistics sector.
- Strong Sales Growth: Palantir reported an impressive 85% revenue growth in Q1, with net income approximately quadrupling year-over-year, showcasing its robust performance in the defense technology sector and projecting a doubling of its U.S. business in the coming year.
- Tesla and SEC Settlement: Tesla CEO Elon Musk reached a settlement with the SEC, agreeing to pay $1.5 million in penalties over allegations of failing to disclose his Twitter stake prior to acquisition, although this minor fine is not expected to have a significant financial impact on the company.
- GLP-1 Market Dynamics: Novo Nordisk's Wegovy pill has rapidly gained tens of thousands of new users within four months of launch, while Eli Lilly's competing Foundayo has seen a more modest uptake, reflecting a shift in market demand for weight-loss medications.
See More
- Market Uncertainty Intensifies: Rising tensions in the Middle East have led to surging oil prices, causing the Dow Jones Industrial Average to drop over 550 points on Monday, marking its worst performance in more than a month, which has left investors concerned about future market sentiment.
- Palantir Surpasses Expectations: Palantir reported an 85% revenue growth in Q1, with net income nearly quadrupling year-over-year, and anticipates its U.S. business will double again next year, raising its full-year free cash flow guidance to $4.2 billion to $4.4 billion, indicating strong market demand and growth potential.
- Tesla Settles with SEC: Tesla CEO Elon Musk has agreed to pay $1.5 million to the SEC to settle allegations of failing to disclose his stake in Twitter before its acquisition, which, if approved by the judge, could alleviate legal risks for Musk and potentially have a positive impact on Tesla's stock price.
- Amazon's New Logistics Initiative: Amazon announced a new initiative allowing external businesses to utilize its supply chain networks for deliveries, which has raised concerns about increased competition with UPS and FedEx, leading to stock declines of over 10% and 9% respectively, reflecting market apprehension about the new competitive landscape.
See More
- Significant Disease Clearance: In the LUCENT-3 study, 63.5% of patients maintained disease clearance after four years of continuous Omvoh treatment, demonstrating the drug's long-term efficacy in ulcerative colitis, which could alter the disease course for patients.
- Elevated Clinical Standards: This study is the first to evaluate disease clearance using a composite measure of symptomatic, endoscopic, and histologic remission, with 61.3% of patients sustaining clearance under the most stringent criteria, indicating Omvoh's superior treatment effects compared to traditional therapies.
- Consistent Safety Profile: Over four years, Omvoh's safety profile remained consistent with known characteristics, with no new safety signals observed; 12% of patients reported serious adverse events, indicating the drug's acceptability for long-term use.
- Future Research Directions: Lilly is advancing combination studies of Omvoh with other therapies to enhance induction efficacy while maintaining long-term remission, further solidifying its leadership position in the field of immunology.
See More










