UK stocks kick off week higher as commodity-linked shares advance By Reuters
Written by Emily J. Thompson, Senior Investment Analyst
Updated: Mar 17 2025
0mins
Should l Buy AZN?
Source: Investing.com
Market Performance: British stocks rose on Monday, with the FTSE 100 index up 0.6% driven by energy and mining shares, while the FTSE 250 gained 0.2%. Key movements included Shell's 1.6% increase and Phoenix Group's 10% jump after positive profit reports.
Economic Outlook: The Bank of England is expected to maintain steady interest rates amid upcoming central bank decisions, while the OECD has downgraded Britain's economic growth forecast for 2025 and 2026 due to global uncertainties.
Trade with 70% Backtested Accuracy
Stop guessing "Should I Buy AZN?" and start using high-conviction signals backed by rigorous historical data.
Sign up today to access powerful investing tools and make smarter, data-driven decisions.
Analyst Views on AZN
Wall Street analysts forecast AZN stock price to rise
14 Analyst Rating
13 Buy
0 Hold
1 Sell
Strong Buy
Current: 204.990
Low
157.61
Averages
213.64
High
252.18
Current: 204.990
Low
157.61
Averages
213.64
High
252.18
About AZN
AstraZeneca PLC is a United Kingdom-based science-led biopharmaceutical company. The Company focuses on the discovery, development, and commercialization of prescription medicines. The Company operates across therapy areas, including Oncology; Cardiovascular, Renal and Metabolism (CVRM); Respiratory and Immunology (R&I); Vaccines and Immune Therapies (V&I), and Rare Disease. In the Oncology area, its key products include Tagrisso, Imfinzi, Calquence, Lynparza, and Enhertu. The key products of CVRM area include Farxiga/Forxiga, Brilinta/Brilique, Crestor, and Lokelma. In the R&I area, the key products are Symbicort, Fasenra, Breztri/Trixeo, and Tezspire. In the V&I Therapies area, the products are Beyfortus and FluMist. The products in the Rare Disease area are Ultomiris, Soliris, Strensiq, and Koselugo. It has about 191 projects in its development pipeline, including 19 new molecular entities (NMEs) in the late-stage pipeline. The Company distributes its products in over 125 countries.
About the author

Emily J. Thompson
Emily J. Thompson, a Chartered Financial Analyst (CFA) with 12 years in investment research, graduated with honors from the Wharton School. Specializing in industrial and technology stocks, she provides in-depth analysis for Intellectia’s earnings and market brief reports.
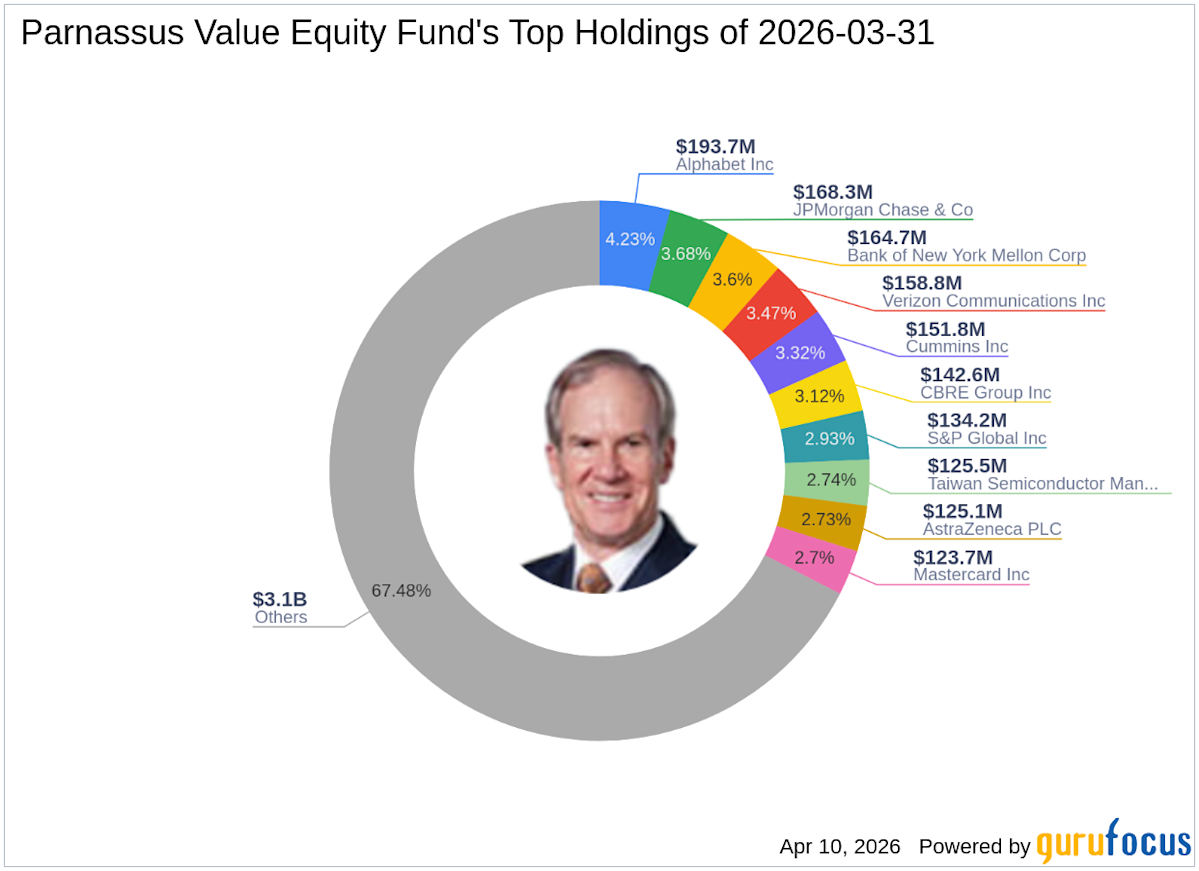
- New Investment Highlights: In Q1 2026, Parnassus Value Equity Fund added 634,492 shares of AstraZeneca (NYSE:AZN), representing 2.73% of the portfolio with a total value of $125.13 million, indicating confidence in the biopharmaceutical sector.
- Key Stock Increases: The fund increased its stake in JPMorgan Chase & Co by 72,858 shares, a 14.59% rise, bringing total holdings to 572,217 shares, reflecting optimism in the financial services industry with a current total value of $168.32 million.
- Strategic Reductions: The fund completely exited its position in AstraZeneca by selling 1,227,628 shares, resulting in a -2.37% impact on the portfolio, showcasing its agility in responding to market dynamics.
- Industry Concentration Analysis: As of Q1 2026, the fund's portfolio included 45 stocks, primarily concentrated in 10 industries such as Financial Services, Technology, and Healthcare, indicating a strategic approach to diversified investments.
See More
- AstraZeneca Sales Outlook: AstraZeneca's shares have risen 16.8% over the past six months, outperforming the 8.6% growth in the biomedical and genetics sector, with expectations to reach $80 billion in total revenues by 2030, despite facing generic competition and pressures on U.S. oncology sales.
- HSBC Strategic Adjustments: HSBC's shares increased by 23% in the last six months, surpassing the 10.5% growth in the foreign banks sector, as its strategic pivot in Asia and asset optimization are expected to support long-term growth, although revenue growth may be impacted in the short term.
- Disney's Profitability: Disney's stock has gained 19.6% over the past year, significantly outpacing the media industry’s 6.1% growth, with its robust IP portfolio and improved profitability in streaming providing sustainable competitive advantages, despite rising programming costs pressuring margins.
- Microcap Performance: Oak Valley Bancorp and BV Financial have seen their shares rise by 25.1% and 23.1% respectively over the past six months, reflecting strong core earnings momentum and liquidity, although they face risks related to concentration in commercial real estate and credit normalization.
See More
- Generic Drug Approval: The FDA announced on Tuesday the approval of the first set of generic products targeting Farxiga, a diabetes therapy developed by AstraZeneca (AZN), indicating increased market competition.
- Market Impact: Farxiga generated $1.7 billion in U.S. sales last year, reflecting a 1% year-over-year decline, yet its market potential remains significant, with the introduction of generics likely to further compress prices.
- Drug Functionality: As a sodium-glucose cotransporter 2 (SGLT2) inhibitor, Farxiga is primarily used to reduce the risk of hospitalization related to heart failure in adults with type 2 diabetes and to improve glycemic control, highlighting its importance in chronic disease management.
- Manufacturers List: Approved generic manufacturers include Indian companies Aurobindo Pharma, Lupin, and Alembic Pharmaceuticals, which will provide more competitive options in the market, potentially impacting AstraZeneca's market share.
See More
- Drug Tariff Agreement: The UK government has reached an agreement with the US that UK-made drugs will be tariff-free for the next three years, significantly boosting UK pharmaceutical exports, which exceed £5 billion annually.
- Cost-Effectiveness Standard Increase: The deal mandates the UK's National Institute for Health and Care Excellence (NICE) to raise its cost-effectiveness threshold by 25%, which will encourage pharmaceutical companies to adjust drug pricing competitively, impacting market supply.
- New Drug Price Increase: The National Health Service (NHS) will pay 25% higher list prices for new prescription drugs, potentially increasing the financial burden on patients while also affecting pharmaceutical companies' profit structures.
- Increased R&D Investment: The UK will raise its drug R&D spending from 0.3% to 0.6% of GDP, a strategic investment aimed at enhancing the UK's competitiveness in the global pharmaceutical industry and fostering innovation and long-term growth.
See More
- Clinical Trial Success: AstraZeneca's antitumor agent Imfinzi demonstrated significant improvement in progression-free survival in the EMERALD-3 Phase 3 trial for hepatocellular carcinoma patients, indicating its potential in treating this cancer type.
- Combination Therapy Evaluation: The trial assessed the efficacy of Imfinzi in combination with CTLA-4 blocker Imjudo, transarterial chemoembolization, and lenvatinib, enrolling 760 patients with unresectable HCC, showcasing the effectiveness of the combination regimen.
- Safety Analysis: An interim analysis revealed that the safety profile of the Imfinzi combination therapy was consistent with the established safety profiles of individual drugs, with no new safety events reported, indicating its clinical safety.
- Regulatory Discussions Ongoing: AstraZeneca is in discussions with global regulatory authorities regarding these positive data while awaiting final results on key secondary endpoints to further confirm the trend in overall survival.
See More
- Significant Trial Results: AstraZeneca's EMERALD-3 Phase III trial demonstrates that the combination of Imfinzi with Imjudo, lenvatinib, and TACE significantly improves progression-free survival compared to TACE alone in patients with unresectable liver cancer, indicating the combination therapy's important clinical significance in enhancing patient outcomes.
- Patient Population Size: By 2026, over 200,000 hepatocellular carcinoma patients are expected to be eligible for embolisation, a standard treatment that blocks blood supply to tumors and delivers chemotherapy directly to the liver, highlighting the market potential for this drug combination.
- Global Trial Scope: The EMERALD-3 trial was conducted across 171 centers in 22 countries, involving 760 patients with unresectable HCC, utilizing a randomized, open-label design that ensures the broad applicability and reliability of the results.
- Future Plans: AstraZeneca plans to present the trial data at an upcoming medical meeting and share it with global regulatory authorities, indicating the company's confidence in the therapy and its potential application in early-stage liver cancer treatment.
See More