Neurocrine Acquires Soleno for $2.9 Billion to Expand Rare Disease Portfolio
Written by Emily J. Thompson, Senior Investment Analyst
Updated: 1 hour ago
0mins
Should l Buy SLNO?
Source: stocktwits
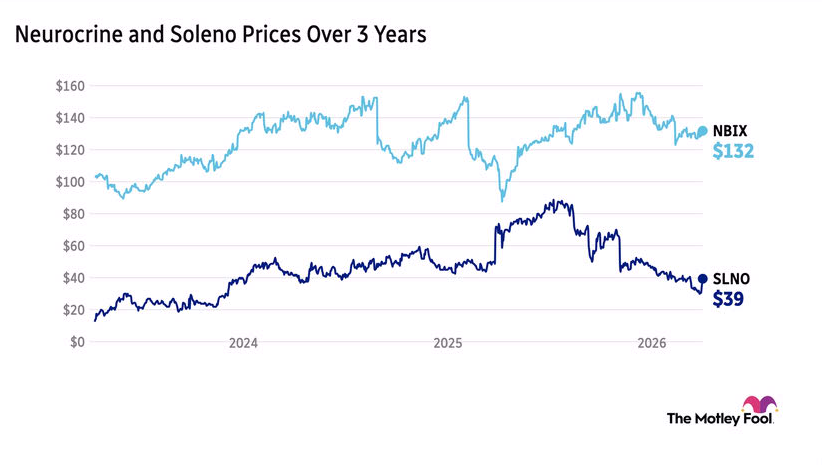
- Acquisition Overview: Neurocrine Biosciences has agreed to acquire Soleno Therapeutics for approximately $2.9 billion, aiming to expand its market presence in rare diseases and endocrinology, highlighting the company's strategic focus on this sector.
- Share Price Premium: The acquisition values Soleno shares at $53 each, representing a notable 34% premium over Friday's closing price, reflecting positive market expectations and recognition of Soleno's future potential.
- Therapy Integration: The acquisition centers on Soleno's therapy VYKAT XR (diazoxide choline), which targets hyperphagia associated with Prader-Willi syndrome, expected to enhance Neurocrine's product offerings in this therapeutic area.
- Market Reaction: Following the announcement, Soleno Therapeutics' stock traded over 39% higher in Monday's premarket, indicating strong investor response and optimism regarding the company's future prospects post-acquisition.
Trade with 70% Backtested Accuracy
Stop guessing "Should I Buy SLNO?" and start using high-conviction signals backed by rigorous historical data.
Sign up today to access powerful investing tools and make smarter, data-driven decisions.
Analyst Views on SLNO
Wall Street analysts forecast SLNO stock price to rise
8 Analyst Rating
8 Buy
0 Hold
0 Sell
Strong Buy
Current: 39.490
Low
75.00
Averages
110.50
High
125.00
Current: 39.490
Low
75.00
Averages
110.50
High
125.00
About SLNO
Soleno Therapeutics, Inc. is a biopharmaceutical company. The Company is focused on developing novel therapeutics for the treatment of rare diseases. The Company’s lead product candidate, diazoxide choline extended-release tablets (DCCR), is for the treatment of Prader-Willi syndrome (PWS) in individuals four years and older who have hyperphagia. DCCR contains diazoxide choline, a potent ATP-sensitive potassium (KATP) channel activator. DCCR tablets consist of the active ingredient diazoxide choline, a choline salt of diazoxide, which is a benzothiadiazine. Its proposed mode of action, with targets in the brain, pancreas and fat tissue, has the potential to broadly impact complex diseases like PWS to reduce appetite, reduce food-seeking, decrease insulin and leptin resistance, and reduce body fat. The Company has Breakthrough Therapy and Fast-Track designations in the United States and Orphan Drug designations in the United States and European Union.
About the author

Emily J. Thompson
Emily J. Thompson, a Chartered Financial Analyst (CFA) with 12 years in investment research, graduated with honors from the Wharton School. Specializing in industrial and technology stocks, she provides in-depth analysis for Intellectia’s earnings and market brief reports.

- Acquisition Talks Progress: Neurocrine Biosciences is in advanced discussions to acquire Soleno Therapeutics, with the deal potentially valued at over $2.5 billion, leading to a 26% surge in premarket trading, reflecting strong market optimism about the transaction.
- Market Opportunity: Should the acquisition proceed, Neurocrine would gain access to Soleno's treatment for Prader-Willi syndrome, which analysts project could generate up to $2.3 billion in peak annual sales, enhancing the strategic appeal of the acquisition.
- Price Target Analysis: The anticipated acquisition price is in the range of $50 to $60 per share, indicating investor confidence in Soleno's future growth potential while providing Neurocrine with an opportunity to enter new markets.
- Warning Signals: Despite the stock surge, GuruFocus has flagged 4 warning signs for Soleno, advising investors to cautiously assess its true value to avoid potential investment risks.
See More
- Lawsuit Background: A securities class action has been filed against Soleno Therapeutics (NASDAQ: SLNO) to represent investors who purchased common stock between March 26 and November 4, 2025, following a disappointing report on DCCR (trademarked as VYKAT™ XR) released on November 5, 2025.
- Stock Price Plunge: The report triggered a 26% drop in Soleno's stock price on the day of release, reflecting strong market concerns about the company's future commercial prospects and prompting Hagens Berman to investigate potential violations of federal securities laws.
- Safety Concerns: The lawsuit alleges that Soleno systematically downplayed and concealed significant safety risks associated with DCCR, including issues of fluid retention in clinical trial participants, which could materially diminish the drug's commercial viability.
- Eroded Market Confidence: Since August 15, 2025, Soleno's stock has fallen nearly 40%, with management acknowledging during the Q3 2025 earnings call that the short seller's report significantly disrupted their launch trajectory, further undermining investor confidence in the company's future.
See More
- Acquisition Overview: Neurocrine Biosciences has agreed to acquire Soleno Therapeutics for approximately $2.9 billion, aiming to expand its market presence in rare diseases and endocrinology, highlighting the company's strategic focus on this sector.
- Share Price Premium: The acquisition values Soleno shares at $53 each, representing a notable 34% premium over Friday's closing price, reflecting positive market expectations and recognition of Soleno's future potential.
- Therapy Integration: The acquisition centers on Soleno's therapy VYKAT XR (diazoxide choline), which targets hyperphagia associated with Prader-Willi syndrome, expected to enhance Neurocrine's product offerings in this therapeutic area.
- Market Reaction: Following the announcement, Soleno Therapeutics' stock traded over 39% higher in Monday's premarket, indicating strong investor response and optimism regarding the company's future prospects post-acquisition.
See More
- Acquisition Talk Boosts Stock: Neurocrine Biosciences is reportedly close to acquiring Soleno Therapeutics for over $2.5 billion, causing Soleno's stock to surge over 30% overnight, with expectations of a share price exceeding $50, reflecting strong market optimism about the deal.
- New Drug Approval Drives Profitability: Soleno's first approved treatment for hyperphagia due to Prader-Willi syndrome has achieved profitability in Q4, marking a significant success in the rapidly growing genetic obesity treatment market.
- Market Rebound Trend: The S&P 500 rose 3.4% and the Nasdaq 4.4% in a trading week shortened by Good Friday, driven by a positive jobs report showing 178,000 new jobs added in March, boosting confidence in economic recovery.
- Coinbase Seeks Banking Charter: Coinbase has received conditional approval to establish a national trust bank, despite regulatory scrutiny, aiming to provide cryptocurrency custody services, indicating its strategic expansion in the fintech sector.
See More
- Acquisition Overview: Neurocrine Biosciences has announced a definitive agreement to acquire Soleno Therapeutics for $53.00 per share in cash, totaling approximately $2.9 billion, significantly enhancing Neurocrine's product portfolio.
- Product Integration Advantage: The acquisition brings Soleno's lead product, VYKAT XR, into Neurocrine's lineup, which is the first FDA-approved treatment for Prader-Willi syndrome, expected to boost the company's market share in this niche area.
- Financial Growth Potential: Since its U.S. launch in 2025, VYKAT XR has generated $190 million in revenue, including $92 million in the fourth quarter alone, indicating strong market demand and growth potential for Neurocrine.
- Strategic Partnership Outlook: Neurocrine's CEO stated that this acquisition will accelerate revenue growth and diversify the product portfolio, laying a solid foundation for long-term revenue growth, particularly within the Prader-Willi syndrome patient community.
See More
- Acquisition Agreement: Neurocrine Biosciences has signed a definitive agreement to acquire Soleno Therapeutics for $53 per share in cash, totaling $2.9 billion, which represents a 34% premium over Soleno's April 2 closing price, indicating strong market confidence in the deal.
- Funding Clarity: The transaction will be financed through cash on hand and a modest amount of debt, with an expected closing within 90 days, reflecting the company's solid financial management and confidence in future growth.
- Portfolio Expansion: The acquisition will add VYKAT XR, the FDA-approved treatment for hyperphagia in Prader-Willi syndrome, to Neurocrine's portfolio, which is projected to generate $190 million in revenue in 2025, further solidifying its position in endocrinology and rare diseases.
- Long-term Growth Potential: This deal will expand Neurocrine's portfolio to three marketed, first-in-class medicines, supporting long-term revenue growth and diversification strategies, thereby enhancing the company's competitiveness in the biopharmaceutical industry.
See More