Celldex Presents Phase 2 Data for Barzolvolimab
Celldex presented additional data from the completed Phase 2 clinical trials of barzolvolimab in chronic spontaneous urticaria and cold urticaria and symptomatic dermographism demonstrating profound improvements in patient quality of life across all measured domains: symptoms and feelings, daily activities, leisure, work and school, personal relationships and treatment. Barzolvolimab is a humanized monoclonal antibody with a completely novel mechanism of action that uniquely targets the root cause of CSU, ColdU and SD-the mast cell. The data were presented at the 2026 Americal Academy of Dermatology Annual Meeting being held March 27 - 31, 2026 in Denver, CO. In the post hoc analysis presented at AAD, the impact of disease on QoL for the subset of patients who received 52 weeks of barzolvolimab therapy and completed treatment with at least well controlled disease at Week 52 and had DLQI data through Week 76 was explored. The vast majority of patients included in this analysis had substantial disease burden at baseline and patients had CSU for a long time. Patients reported that their CSU had a very large negative impact on their QoL. At Week 52, 94% of patients with well controlled disease at Week 52 also had a DLQI of 0/1, indicating their disease had no impact on their QoL . Barzolvolimab treatment led to prolonged, off-treatment enhanced QoL seven months after the last dose of barzolvolimab. At baseline, patients reported their CSU significantly impacted their QoL across all 6 DLQI domains. Profound improvements were observed across all 6 domains at both Weeks 52 and 76. 76% of patients with well controlled disease at Week 52 also reported that the disease had small to no impact on their QoL at Week 76. The majority of patients with well controlled disease at Week 52 achieved DLQI domain scores of 0/1 at Week 76. This sustained off-treatment improvement in QoL was observed despite barzolvolimab clearance and normalization of tryptase, suggesting disease modification and supports barzolvolimab's significant potential to become a transformative treatment option for patients suffering from CSU.
Trade with 70% Backtested Accuracy
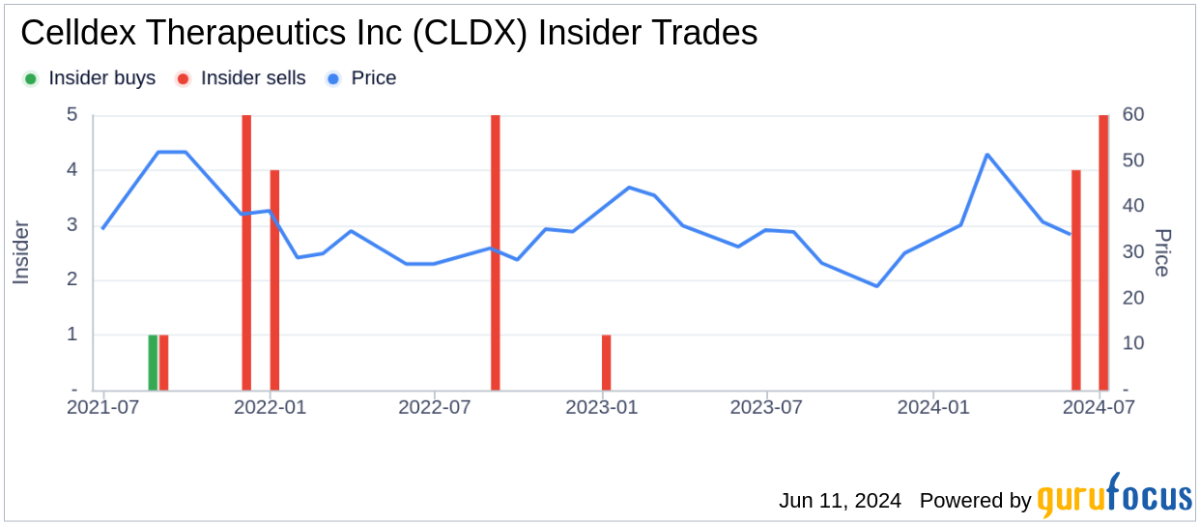
Analyst Views on CLDX
About CLDX
About the author

- Offering Size: Celldex Therapeutics successfully closed its public offering of 11,896,750 shares of common stock, which included the full exercise of the underwriters' option to purchase an additional 1,551,750 shares at a public offering price of $29.00 per share, indicating strong market demand for its stock.
- Funding Amount: The offering generated approximately $345 million in gross proceeds for Celldex, excluding underwriting discounts and other related expenses, with the funds earmarked to advance its biotechnology research projects, thereby enhancing the company's competitive edge in the clinical stage.
- Underwriter Lineup: The offering was managed by Leerink Partners, TD Cowen, Guggenheim Securities, and Cantor as joint bookrunning managers, with LifeSci Capital and H.C. Wainwright & Co. serving as co-lead managers, reflecting robust market support for the transaction.
- Registration Statement: The securities were offered under a shelf registration statement on Form S-3 filed with the SEC, demonstrating Celldex's commitment to compliance and laying the groundwork for future financing activities.
- Public Offering Pricing: Celldex Therapeutics has priced an underwritten public offering of approximately 10.3 million shares at $29.00 per share, expecting gross proceeds of about $300 million, which underscores the company's ability to raise capital in the market.
- Additional Share Option: The underwriters have a 30-day option to purchase up to an additional 1.55 million shares, providing the company with flexibility for further financing and potentially enhancing market confidence.
- Clear Use of Proceeds: The funds raised will support the commercial readiness and potential U.S. launch of barzolvolimab for chronic spontaneous urticaria, if approved, alongside clinical and preclinical development of product candidates, indicating the company's focus on new product initiatives.
- Stock Price Fluctuation: Following the public offering announcement, Celldex's stock price dropped about 3.5% during pre-market trading on Thursday, reflecting a cautious market reaction to the financing news, which may impact investor confidence.
- Public Offering Pricing: Celldex Therapeutics has priced its public offering of 10.345 million shares at $29 each, expecting gross proceeds of $300 million, although the stock fell 3.45% in after-hours trading to $30.18, indicating cautious market sentiment towards the offering.
- Use of Proceeds: The net proceeds from this offering will be combined with existing cash and equivalents to fund ongoing commercial readiness and the potential launch of Barzolvolimab, if approved, while also supporting preclinical development of product candidates, demonstrating the company's commitment to drug development.
- Financial Condition Analysis: Celldex anticipates a wider net loss of $258.76 million for 2025, translating to a loss of $3.90 per share, compared to a loss of $157.86 million the previous year, reflecting high costs associated with R&D and market promotion.
- Declining Revenue Trend: The company's total revenue has decreased from $7.02 million in the prior year to $1.55 million, primarily due to reduced services under agreements with Rockefeller University, indicating challenges in revenue diversification.
- Offering Size: Celldex Therapeutics announced a public offering of 10,345,000 shares at $29.00 per share, expected to raise approximately $300 million, reflecting strong market demand and investor confidence in the company.
- Underwriter Selection: The offering is managed by Leerink Partners, TD Cowen, Guggenheim Securities, and Cantor, indicating the company's focus on reputable underwriters to ensure successful capital raising and market impact.
- Use of Proceeds: Celldex intends to utilize the net proceeds from this offering to fund commercial readiness and launch of barzolvolimab, along with ongoing clinical and preclinical development, thereby enhancing its product pipeline and competitive positioning.
- Compliance and Transparency: The offering is conducted under SEC's S-3 registration statement, ensuring compliance and transparency, which further builds investor trust in the company's governance and financial health.
- Offering Size: Celldex Therapeutics announced a public offering of 10,345,000 shares at a price of $29.00 per share, expecting to raise approximately $300 million in gross proceeds, which will significantly enhance the company's financial strength to support future R&D and commercialization activities.
- Underwriter Selection: The offering is managed by Leerink Partners, TD Cowen, Guggenheim Securities, and Cantor, reflecting strong market confidence in Celldex while providing robust capital market support for the company.
- Planned Use of Proceeds: Celldex intends to use the net proceeds from this offering to fund commercial readiness and launch of barzolvolimab, along with ongoing clinical and preclinical development, indicating the company's proactive strategy in drug development and market entry.
- Compliance and Transparency: The offering is made under a previously filed S-3 registration statement, ensuring compliance and transparency, which further enhances investor trust in Celldex and lays a solid foundation for future capital operations.
- Public Offering Launch: Celldex Therapeutics has announced the commencement of an underwritten public offering to sell shares of its common stock, with an expected 30-day option for underwriters to purchase an additional 15% of the shares to enhance financing flexibility.
- Clear Use of Proceeds: The net proceeds from this offering will be combined with existing cash and marketable securities, primarily aimed at funding the commercial readiness and launch of barzolvolimab for the treatment of chronic spontaneous urticaria, pending approval.
- Ongoing R&D Advancement: Celldex plans to utilize the offering proceeds to continue the clinical and preclinical development of its product candidates, particularly focusing on the current and future development of barzolvolimab, while also expanding its bispecific antibody platform and clinical candidates.
- Strategic Corporate Positioning: This financing not only supports new drug development but also provides funding for the development of other clinical pipeline products, demonstrating Celldex's proactive positioning for future market opportunities.