Amgen's Tepezza Treatment Achieves Phase 3 Success
Written by Emily J. Thompson, Senior Investment Analyst
Updated: 2 hours ago
0mins
Should l Buy AMGN?
Source: seekingalpha
- Clinical Trial Results: Amgen's Tepezza (teprotumumab) has successfully met both primary and secondary endpoints in its phase 3 trial for the subcutaneous version, marking a significant advancement in the treatment of thyroid eye disease.
- Efficacy Data: Patients treated with Tepezza OBI demonstrated a proptosis response rate of approximately 77% over 24 weeks, compared to only 20% in the placebo group, indicating a substantial therapeutic advantage.
- Secondary Endpoint Improvements: The mean proptosis reduction at week 24 was -3.17 mm for the Tepezza group versus -0.80 mm for the placebo, reinforcing the treatment's effectiveness and its potential market impact.
- Quality of Life Enhancement: Other secondary endpoints showed significant improvements, including overall responder rates and changes in diplopia and Graves' Ophthalmopathy Quality of Life scores, suggesting that Tepezza not only alleviates symptoms but also enhances overall patient quality of life.
Trade with 70% Backtested Accuracy
Stop guessing "Should I Buy AMGN?" and start using high-conviction signals backed by rigorous historical data.
Sign up today to access powerful investing tools and make smarter, data-driven decisions.
Analyst Views on AMGN
Wall Street analysts forecast AMGN stock price to rise
24 Analyst Rating
14 Buy
9 Hold
1 Sell
Moderate Buy
Current: 347.940
Low
280.00
Averages
363.10
High
425.00
Current: 347.940
Low
280.00
Averages
363.10
High
425.00
About AMGN
Amgen Inc. is a biotechnology company. It discovers, develops, manufactures and delivers medicines for the toughest diseases. It focuses on areas of high unmet medical need and leverages its expertise to strive for solutions that improve people’s lives. It operates in the human therapeutics segment. Its marketed products portfolio includes EPOGEN (epoetin alfa); Aranesp (darbepoetin alfa); Parsabiv (etelcalcetide); Neulasta (pegfilgrastim); KANJINTI (trastuzumab-anns); Otezla; BLINCYTO (blinatumomab); ACTIMMUNE (interferon gamma-1b); Neulasta (pegfilgrastim); Sensipar/Mimpara (cinacalcet); Prolia (denosumab); ENBREL; QUINSAIR (levofloxacin); Repatha (evolocumab) and others. It markets ENBREL, a tumor necrosis factor blocker, in the United States and Canada. It markets Otezla, a small molecule that inhibits phosphodiesterase 4, in many countries around the world. It markets Repatha, a proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor, in many countries around the world.
About the author

Emily J. Thompson
Emily J. Thompson, a Chartered Financial Analyst (CFA) with 12 years in investment research, graduated with honors from the Wharton School. Specializing in industrial and technology stocks, she provides in-depth analysis for Intellectia’s earnings and market brief reports.
- Clinical Trial Success: Amgen's TEPEZZA demonstrated a 77% proptosis response rate in its Phase 3 trial for moderate-to-severe active Thyroid Eye Disease, significantly outperforming the 19.6% response in the placebo group, indicating substantial therapeutic efficacy for this rare autoimmune condition.
- Consistent Safety Profile: The overall safety results were consistent with TEPEZZA's known safety profile, suggesting that the new subcutaneous administration method does not introduce significant safety concerns, thereby enhancing market acceptance and patient confidence.
- Innovative Delivery Method: TEPEZZA's subcutaneous injection provides IV-level efficacy, expanding its potential application among Thyroid Eye Disease patients and likely attracting more patients to this delivery method.
- Stable Market Performance: Amgen's stock has fluctuated between $261.43 and $391.29 over the past year, currently trading at $344.33, down 1.04%, yet overall performance reflects market optimism regarding its new treatment options.
See More
- Clinical Trial Results: Amgen's Tepezza (teprotumumab) has successfully met both primary and secondary endpoints in its phase 3 trial for the subcutaneous version, marking a significant advancement in the treatment of thyroid eye disease.
- Efficacy Data: Patients treated with Tepezza OBI demonstrated a proptosis response rate of approximately 77% over 24 weeks, compared to only 20% in the placebo group, indicating a substantial therapeutic advantage.
- Secondary Endpoint Improvements: The mean proptosis reduction at week 24 was -3.17 mm for the Tepezza group versus -0.80 mm for the placebo, reinforcing the treatment's effectiveness and its potential market impact.
- Quality of Life Enhancement: Other secondary endpoints showed significant improvements, including overall responder rates and changes in diplopia and Graves' Ophthalmopathy Quality of Life scores, suggesting that Tepezza not only alleviates symptoms but also enhances overall patient quality of life.
See More

- Clinical Trial Success: Amgen's TEPEZZA OBI demonstrated a 77% proptosis improvement rate in Thyroid Eye Disease (TED) patients, significantly surpassing the 19.6% in the placebo group (p<0.0001), indicating further validation of the drug's efficacy in treating TED.
- Significant Symptom Improvement: Over the 24-week trial, the average proptosis reduction was 3.17 mm in the TEPEZZA OBI group compared to only 0.80 mm in the placebo group, showcasing the potential of TEPEZZA OBI to enhance patient quality of life and likely attracting more patients to this treatment option.
- Good Safety and Tolerability: The safety profile of TEPEZZA OBI was consistent with that of TEPEZZA IV, with mild to moderate injection site reactions observed in some patients but no treatment interruptions, indicating its acceptability in clinical use.
- Future Market Potential: The success of TEPEZZA OBI lays a foundation for Amgen's market expansion in the Thyroid Eye Disease space, expected to attract more patients and enhance the company's competitiveness in the biopharmaceutical market, particularly by offering a more convenient administration method.
See More
- Clinical Trial Success: Amgen's TEPEZZA achieved a statistically significant 77% proptosis response rate in its Phase 3 trial for moderate-to-severe active Thyroid Eye Disease, demonstrating both efficacy and clinical relevance over the 24-week period.
- Innovative Delivery Method: The subcutaneous administration of TEPEZZA provides comparable efficacy to intravenous delivery, enhancing patient convenience and potentially attracting a larger patient base, thereby expanding market share.
- Safety and Tolerability: The trial results indicated that TEPEZZA's safety profile aligns with that of the intravenous formulation, with mild injection site reactions observed but no treatment interruptions, suggesting good tolerability among patients.
- Future Outlook: The success of TEPEZZA not only solidifies its leading position in the treatment of Thyroid Eye Disease but also lays a strong foundation for Amgen's further development in the biopharmaceutical sector, with complete results expected to be presented at an upcoming medical congress.
See More
- AbbVie's Growth Potential: AbbVie expects combined sales of its immunology drugs Skyrizi and Rinvoq to exceed $31 billion by 2026, a year ahead of schedule, demonstrating the company's strong resilience during economic downturns and long-term growth potential.
- Botox's Market Position: AbbVie's Botox product is anticipated to continue generating stable revenue due to its complex molecular structure and strong brand influence, further solidifying its leadership position in the market.
- Amgen's Product Diversity: Despite facing patent expiration challenges, Amgen's products like Tezspire and Repatha continue to drive sales growth, showcasing the company's stability during tough economic times.
- New Drug Development Outlook: Amgen's MariTide, currently in phase 3 trials, could capture a significant share of the rapidly growing weight loss market if successful, enhancing the attractiveness of its product portfolio.
See More
- Market Volatility: On April 2, 2026, U.S. stocks opened lower before recovering slightly, with the Dow Jones Industrial Average struggling to find direction for most of the morning, reflecting market uncertainty.
- Tesla Delivery Miss: Tesla reported first-quarter deliveries of 358,023 vehicles, falling short of Wall Street's expectation of 366,000, resulting in a more than 5% drop in its stock price, which negatively impacted overall market sentiment.
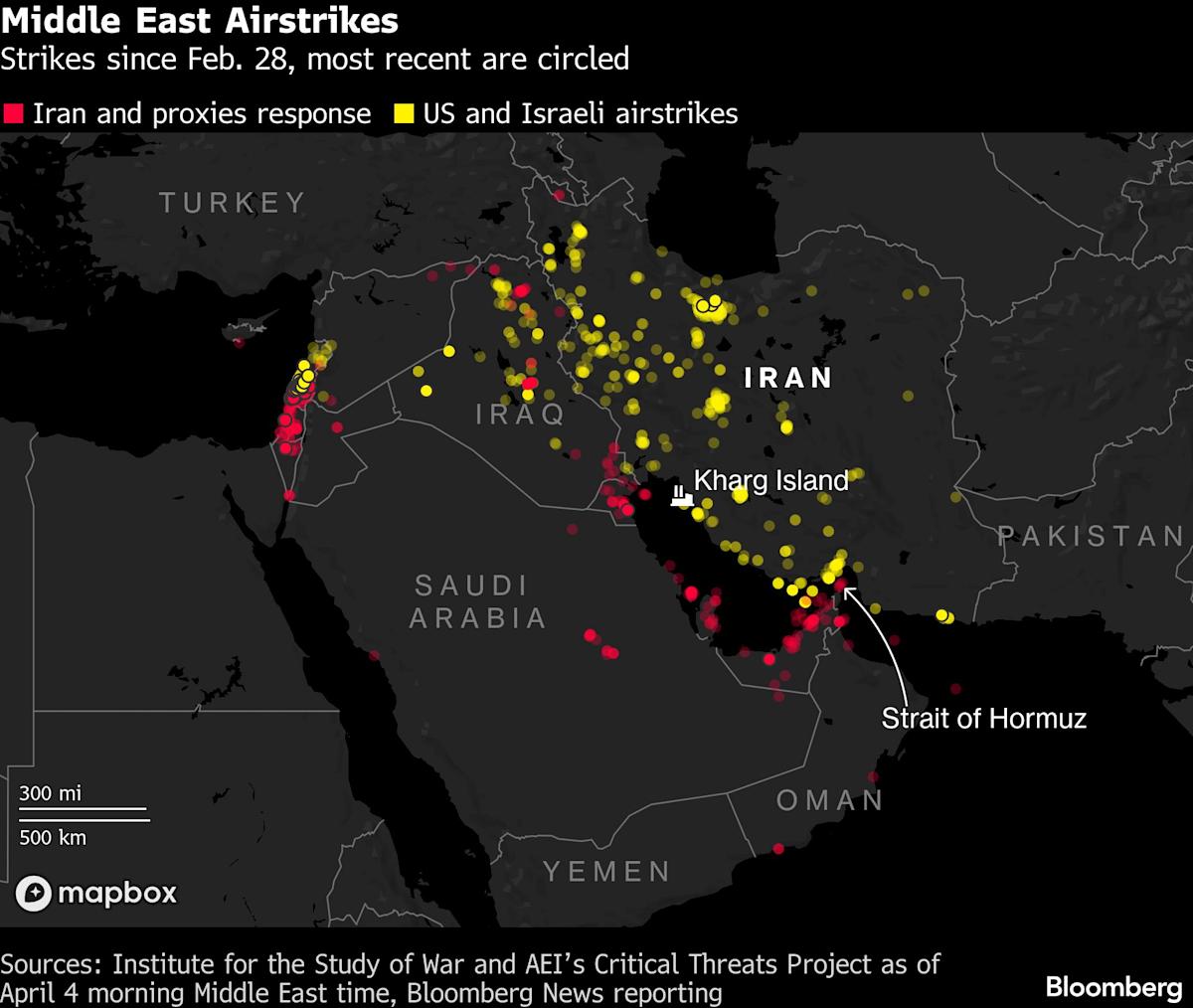
- Surge in Oil Prices: Crude oil prices surged approximately 10% on Thursday due to ongoing tensions in Iran, with market participants anxious about supply disruptions in the Strait of Hormuz, although reports indicated Iran and Oman are working to restore traffic through the strait.
- Healthcare Stocks React: The Trump administration's plans to impose tariffs of up to 100% on certain imported pharmaceuticals led to a roughly 2% decline in healthcare stocks, including major players like Eli Lilly and Amgen, highlighting the market's sensitivity to policy changes.
See More