Launch of HemoSphere Stream™ Module Enhances Noninvasive Blood Pressure Monitoring
Written by Emily J. Thompson, Senior Investment Analyst
Updated: 2 days ago
0mins
Should l Buy BDX?
Source: PRnewswire
- Technological Innovation: BD's launch of the HemoSphere Stream™ Module enables clinicians to obtain continuous, noninvasive arterial waveform data in real-time through compatible multiparameter monitors, significantly enhancing blood pressure monitoring capabilities, especially for patients who do not require invasive arterial lines.
- Clinical Application: The module easily mounts to IV poles without taking up additional space in clinical settings when used with the VitaWave™ Plus finger cuff, thereby improving monitoring efficiency in hospitals and helping physicians quickly identify hemodynamic changes.
- Safety Enhancement: By providing real-time blood pressure trends, the HemoSphere Stream™ Module supports the Anesthesia Patient Safety Foundation's recommendations, facilitating earlier detection and treatment of intraoperative hypotension, which reduces the risk of major complications after surgery.
- Market Potential: As a leading global medical technology company, BD further solidifies its market position in the medical monitoring field with this innovative product, which is expected to drive continued growth in global healthcare safety and efficiency.
Trade with 70% Backtested Accuracy
Stop guessing "Should I Buy BDX?" and start using high-conviction signals backed by rigorous historical data.
Sign up today to access powerful investing tools and make smarter, data-driven decisions.
Analyst Views on BDX
Wall Street analysts forecast BDX stock price to rise
11 Analyst Rating
5 Buy
6 Hold
0 Sell
Moderate Buy
Current: 154.960
Low
157.00
Averages
192.64
High
232.00
Current: 154.960
Low
157.00
Averages
192.64
High
232.00
About BDX
Becton, Dickinson and Company is a global medical technology company. The Company is engaged in the development, manufacture and sale of a broad range of medical supplies, devices, laboratory equipment and diagnostic products used by healthcare institutions, physicians, life science researchers, clinical laboratories, and more. It operates through four segments: BD Medical Essentials, BD Connected Care, BD BioPharma Systems and BD Interventional. BD Medical Essentials segment includes medication delivery solutions and specimen management. BD Connected Care segment includes medication management solutions and advanced patient monitoring. BD Interventional segment includes urology and critical care, peripheral intervention and surgery.
About the author

Emily J. Thompson
Emily J. Thompson, a Chartered Financial Analyst (CFA) with 12 years in investment research, graduated with honors from the Wharton School. Specializing in industrial and technology stocks, she provides in-depth analysis for Intellectia’s earnings and market brief reports.
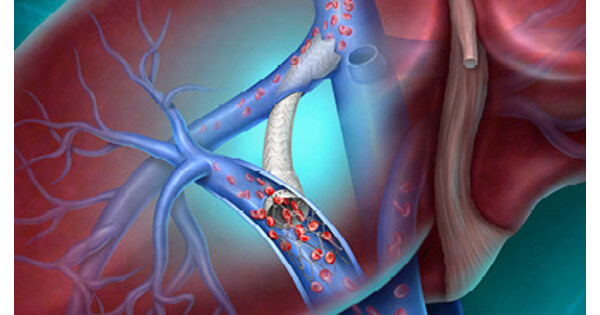
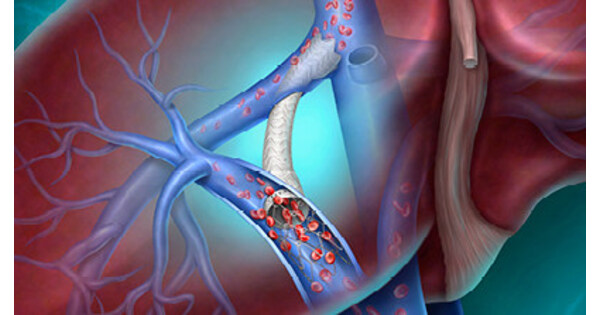
- CE Mark Announcement: BD has announced that the Liverty™ TIPS Stent Graft has received CE Mark approval, aimed at providing innovative treatment options for patients with cirrhosis and portal hypertension, marking a significant advancement in the company's interventional therapies and expected to enhance patient outcomes and quality of life.
- ARCH Clinical Trial Data: Safety and efficacy data for the Liverty™ TIPS Stent Graft will be presented for the first time at the Society of Interventional Radiology Annual Scientific Meeting on April 12, 2026, offering a unique perspective from a global prospective study that could drive broader clinical adoption of the device.
- Technological Innovation: The stent features an adjustable inner diameter of 6-10 mm and a variety of covered lengths, utilizing a self-expanding nitinol frame and dual-layer ePTFE encapsulation, designed to enhance treatment personalization and safety, catering to the physiological needs of diverse patients.
- Global Health Challenge: With approximately 58 million people worldwide suffering from cirrhosis, the introduction of the Liverty™ TIPS Stent Graft not only provides new treatment options but also has the potential to alleviate severe complications arising from portal hypertension, thereby improving overall public health outcomes.
See More
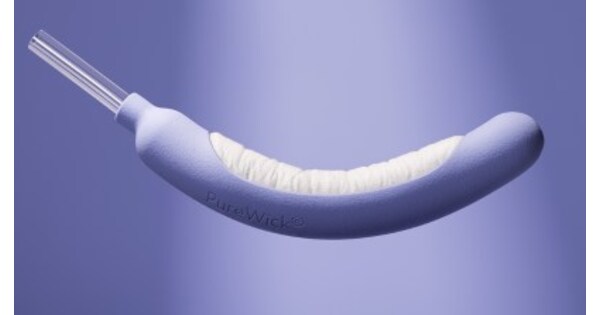
- Clinical Study Findings: A four-week randomized trial involving 165 women revealed that the BD PureWick® Flex Urine Collection System significantly improved sleep quality, demonstrating its effectiveness and comfort in non-invasive urinary management.
- Enhanced User Experience: Participants highlighted the system's ease of use and non-invasive design, allowing them to stay dry overnight and reducing the need for frequent bathroom trips, thereby lowering fall risks, particularly for women aged 65 and older.
- Quality of Life Improvement: The study indicated that women using the system experienced significant enhancements in comfort and confidence, suggesting that the product not only alleviates the daily burden of incontinence but also promotes independence and dignity among users.
- Broad Market Potential: BD's research showcases its innovative capabilities in the non-invasive urinary management device sector, which is expected to drive widespread adoption of the PureWick® system in home care markets, addressing the growing demand.
See More
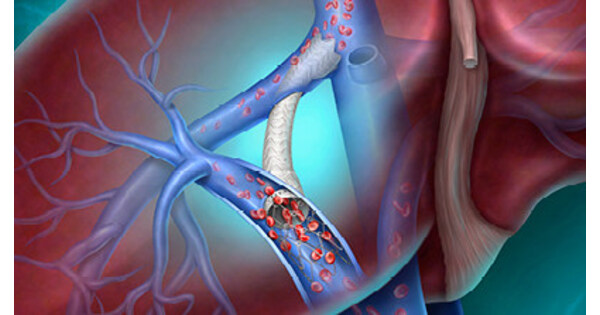
- CE Certification Progress: BD has applied for CE certification for the Liverty™ TIPS Stent, aiming to innovatively advance portal hypertension treatment across the EU, which is expected to significantly enhance treatment options for cirrhosis patients and strengthen the company's competitiveness in the medical device market.
- Clinical Study Data: Safety and efficacy data for the Liverty™ TIPS Stent will be presented for the first time at the Society of Interventional Radiology annual meeting on April 12, 2026, marking BD's latest advancement in interventional therapies for complex liver diseases and potentially attracting more healthcare institutions' interest.
- Personalized Treatment Design: The stent features an adjustable diameter (6 to 10 mm) and a variety of length options, designed to provide personalized treatment plans based on patients' physiological characteristics, thereby improving treatment safety and effectiveness while meeting market demand for flexible treatment solutions.
- Global Health Challenge: According to the Global Burden of Disease study, approximately 58 million people are affected by liver cirrhosis, and the launch of the Liverty™ TIPS Stent will help alleviate severe complications caused by portal hypertension, further solidifying BD's leadership position in the global healthcare sector.
See More
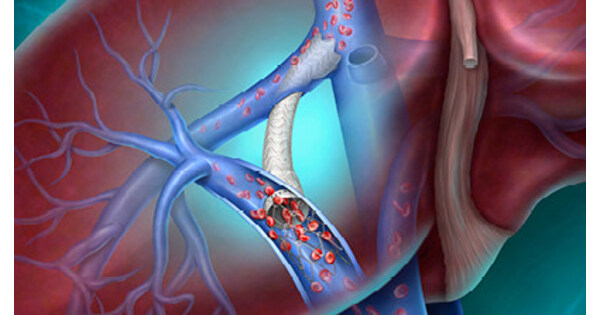
- Innovative Medical Device: BD's Liverty™ TIPS stent has received CE marking, aimed at providing safer and more effective treatment options for patients with advanced liver disease, particularly for complications related to cirrhosis and portal hypertension, marking the company's latest advancement in interventional therapies.
- Clinical Trial Data: The safety and efficacy of Liverty™ TIPS will be presented for the first time at the Society of Interventional Radiology annual meeting on April 12, 2026, which is expected to provide crucial clinical evidence for physicians to optimize personalized treatment plans for patients.
- Market Demand Response: With approximately 58 million people affected by liver cirrhosis globally, the launch of Liverty™ TIPS not only meets the needs of this vast market but may also enhance BD's competitive position by improving long-term treatment outcomes for patients.
- Technical Advantages: The stent features an adjustable diameter (6 to 10 mm) and a flexible self-expanding design, allowing for better adaptation to the unique physiological characteristics of patients, thereby increasing the safety and efficacy of treatments and further solidifying BD's leadership in the medical technology sector.
See More
- Innovative Treatment Solution: BD has announced the CE mark application for the Liverty™ TIPS stent, aimed at providing broader treatment options for patients with cirrhosis and portal hypertension complications, which is expected to significantly enhance patient outcomes and quality of life.
- Clinical Data Release: Safety and efficacy data for the Liverty™ TIPS stent will be presented for the first time at the SIR Annual Scientific Meeting on April 12, 2026, marking the official launch of the product in the European market and potentially driving BD's market share growth in the medical device sector.
- Technological Advantages: The stent features an adjustable diameter (6 to 10 mm) and a range of length options, utilizing a self-expanding nitinol structure designed to enhance physician flexibility and precision during treatment, thereby improving the personalized treatment experience for patients.
- Global Health Challenge: With approximately 58 million people affected by liver cirrhosis worldwide, the introduction of the Liverty™ TIPS stent not only provides new treatment options for patients but may also play a crucial role in addressing global health challenges, further solidifying BD's leadership position in the medical technology field.
See More
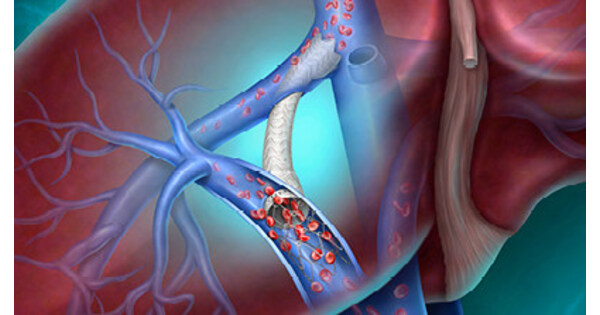
- Innovative Medical Device: BD's Liverty™ TIPS Stent Graft has received CE Mark approval, aimed at providing safer and more effective treatment options for patients with cirrhosis and portal hypertension, marking the company's latest advancement in interventional therapies.
- Clinical Trial Data Release: Safety and effectiveness data from the ARCH clinical trial will be presented for the first time at the Society of Interventional Radiology Annual Scientific Meeting on April 12, 2026, which is expected to provide significant support for the clinical application of this device.
- Personalized Treatment Solutions: The stent features an adjustable inner diameter of 6-10 mm and a broad range of covered lengths, allowing for individualized management based on patients' unique physiological characteristics, thereby enhancing treatment efficacy and long-term outcome potential.
- Global Health Challenge: With approximately 58 million people worldwide suffering from cirrhosis, the introduction of the Liverty™ TIPS Stent Graft will help alleviate severe complications arising from portal hypertension, ultimately improving patients' quality of life.
See More