Hutchmed Reports SACHI Phase III Trial Results
Hutchmed (HCM) reported that results from the SACHI Phase III trial were published in The Lancet. SACHI is a Phase III study of the savolitinib and osimertinib combination for the treatment of patients with locally advanced or metastatic epidermal growth factor receptor mutation-positive non-small cell lung cancer with MET amplification after disease progression on first-line EGFR tyrosine kinase inhibitor therapy. Savolitinib is an oral, potent and highly selective MET TKI being jointly developed by AstraZeneca (AZN) and Hutchmed and commercialized by AstraZeneca. Osimertinib is a third-generation, irreversible EGFR TKI. Based on interim data from SACHI, the savolitinib and osimertinib combination was granted regulatory approval in China in June 2025.
Trade with 70% Backtested Accuracy
Analyst Views on AZN
About AZN
About the author

- Trial Success: AstraZeneca announced a successful late-stage trial of its antibody therapy tozorakimab in the Phase 3 MIRANDA trial for chronic obstructive pulmonary disease (COPD), significantly reducing the annualized rate of moderate-to-severe exacerbations with both statistical and clinical significance.
- Broad Applicability: The therapy demonstrated efficacy across both former and current smokers, indicating its effectiveness regardless of lung function severity and blood eosinophil counts, thereby enhancing its market potential.
- Study Design: The 52-week study evaluated tozorakimab 300 mg administered biweekly against a placebo, aiming to assess its additional efficacy on top of standard inhaled therapy, further validating its clinical application value.
- Favorable Safety Profile: AstraZeneca noted that tozorakimab was generally well tolerated with a safety profile consistent with prior trials, and plans to submit the results to global regulators and present them at future medical events, facilitating its market introduction.
- Clinical Trial Results: AstraZeneca's IL-33-targeting biologic tozorakimab demonstrated significant efficacy in the Phase III MIRANDA trial, showing a meaningful reduction in the annualized rate of moderate-to-severe COPD exacerbations, particularly among former smokers, indicating its potential first-in-class status in COPD treatment.
- Patient Recruitment: The MIRANDA trial enrolled 1,454 patients, all of whom received tozorakimab 300mg alongside standard care, with results highlighting the drug's important clinical benefits for patients still experiencing exacerbations, thus addressing a critical need in COPD management.
- Safety Profile: Tozorakimab was generally well tolerated with a favorable safety profile consistent with previous trials, providing strong support for its future regulatory approval and the potential to offer new treatment options for COPD patients.
- Market Potential: With COPD being the third leading cause of death globally, affecting nearly 400 million people, the success of tozorakimab could significantly enhance patient quality of life and further bolster AstraZeneca's leadership position in the respiratory therapeutics market.
- Share Increase: On April 14, 2026, Pictet North America Advisors SA disclosed an increase of 3,529 shares in MercadoLibre, with an estimated transaction value of $6.81 million, indicating confidence in the company's future growth prospects.
- Stake Growth: Following this purchase, Pictet's stake in MercadoLibre rose to 2.21%, reflecting its emphasis on the company within its portfolio, particularly as MercadoLibre's market capitalization reached $92.87 billion.
- Financial Performance: Despite achieving a 44% revenue growth in 2025, MercadoLibre's net income only increased by 5% due to heightened competition and rising non-performing loans, indicating some profitability pressures.
- Market Outlook: Although facing short-term challenges, MercadoLibre is expected to maintain long-term growth potential due to its market leadership in Latin America and robust revenue growth, suggesting that Pictet's decision to increase its holdings may be based on confidence in this outlook.
- Clinical Trial Results: AbbVie's March 9 announcement of ABBV-295's clinical trial results indicates that patients receiving weekly treatment lost an average of 7.75% to 9.79% of their weight over 12 weeks, while those on biweekly or monthly regimens lost between 7.86% and 9.73% over 13 weeks, suggesting strong potential for the drug in the weight loss market.
- Competitive Market Pressure: Despite the promising initial data for ABBV-295, AbbVie faces intense competition in the weight loss market from companies like Eli Lilly, which have several late-stage obesity drugs, indicating that the market will become increasingly crowded before AbbVie can launch its product.
- Core Business Stability: AbbVie's primary therapeutic area remains immunology, with projected sales for Skyrizi and Rinvoq exceeding $31 billion in 2023, significantly surpassing Humira's peak sales, demonstrating the company's robust performance and growth potential in this sector.
- Optimistic Pipeline Outlook: Even if ABBV-295 fails in clinical trials, AbbVie has a deep pipeline of investigational products, including the ongoing ABBV-383 cancer treatment, which is expected to provide significant support for the company's future financial performance and further solidify its leadership in the pharmaceutical industry.
- Market Surge: According to Markets and Markets, the GLP-1 agonist market grew from $53.74 billion in 2024 to $64.42 billion in 2025, with projections reaching $170.75 billion by 2033, indicating a robust demand for obesity and diabetes treatments.
- New Drug Approval: In 2025, Novo Nordisk's Wegovy received FDA approval as the first oral GLP-1 weight-loss medication, marking a significant breakthrough in treatment options, particularly for those with trypanophobia, which is expected to further drive market demand.
- Top-Selling Drugs: In 2025, Mounjaro led the GLP-1 drug market with $22.97 billion in sales, followed by Ozempic and Zepbound with $20 billion and $13.54 billion, respectively, showcasing the strong appeal of GLP-1 therapies globally.
- Patent Protection and Competition: While Ozempic and Wegovy are protected by patents in the U.S. until around 2032, generics have already launched in India and are expected in China, intensifying market competition and potentially impacting the market share of original branded drugs.
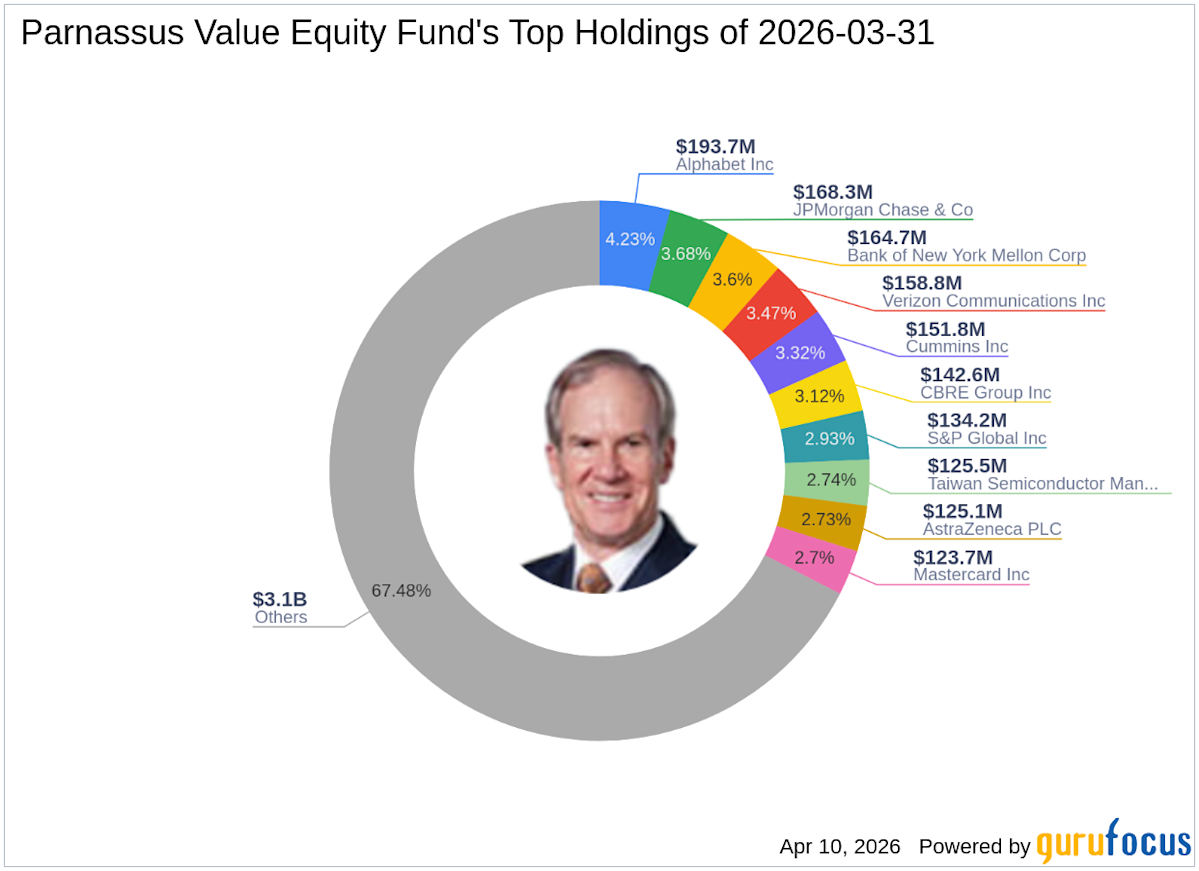
- New Investment Highlights: In Q1 2026, Parnassus Value Equity Fund added 634,492 shares of AstraZeneca (NYSE:AZN), representing 2.73% of the portfolio with a total value of $125.13 million, indicating confidence in the biopharmaceutical sector.
- Key Stock Increases: The fund increased its stake in JPMorgan Chase & Co by 72,858 shares, a 14.59% rise, bringing total holdings to 572,217 shares, reflecting optimism in the financial services industry with a current total value of $168.32 million.
- Strategic Reductions: The fund completely exited its position in AstraZeneca by selling 1,227,628 shares, resulting in a -2.37% impact on the portfolio, showcasing its agility in responding to market dynamics.
- Industry Concentration Analysis: As of Q1 2026, the fund's portfolio included 45 stocks, primarily concentrated in 10 industries such as Financial Services, Technology, and Healthcare, indicating a strategic approach to diversified investments.