FDA Approves Biogen's New High-Dose Spinraza Regimen
Written by Emily J. Thompson, Senior Investment Analyst
Updated: 5 hours ago
0mins
Should l Buy BIIB?
Source: seekingalpha
- New Dosage Regimen: The US FDA has approved Biogen's new high-dose regimen for Spinraza, consisting of 50 mg/5 mL and 28 mg/5 mL doses, significantly enhancing treatment flexibility and efficacy.
- Accelerated Loading Phase: The new regimen allows individuals new to treatment to receive two 50 mg high-dose injections 14 days apart, followed by 28 mg maintenance doses every four months, thereby accelerating patient treatment progress.
- Transition for Low-Dose Patients: Patients switching from the low-dose regimen will follow their current dosing schedule every four months after a single high-dose loading phase, ensuring continuity and stability in their treatment.
- Clinical Study Support: The approval is supported by results from the phase 2/3 DEVOTE study, demonstrating significant advantages in treatment outcomes with the new dosing regimen, further solidifying Biogen's market position in spinal muscular atrophy.
Trade with 70% Backtested Accuracy
Stop guessing "Should I Buy BIIB?" and start using high-conviction signals backed by rigorous historical data.
Sign up today to access powerful investing tools and make smarter, data-driven decisions.
Analyst Views on BIIB
Wall Street analysts forecast BIIB stock price to rise
26 Analyst Rating
11 Buy
14 Hold
1 Sell
Moderate Buy
Current: 183.840
Low
143.00
Averages
204.45
High
246.00
Current: 183.840
Low
143.00
Averages
204.45
High
246.00
About BIIB
Biogen Inc. is a global biopharmaceutical company. The Company is focused on discovering, developing, and delivering advanced therapies for people living with serious and complex diseases worldwide. It operates a portfolio of medicines to treat multiple sclerosis (MS), spinal muscular atrophy (SMA), Alzheimer's disease, and amyotrophic lateral sclerosis (ALS). It is focused on advancing its pipeline in neurology, specialized immunology, and rare diseases. Its marketed products include TECFIDERA, VUMERITY, AVONEX, PLEGRIDY, TYSABRI and FAMPYRA for the treatment of MS; SPINRAZA for the treatment of SMA; SKYCLARYS for the treatment of Friedreich's Ataxia; QALSODY for the treatment of ALS, and FUMADERM for the treatment of severe plaque psoriasis. It also collaborations with Eisai on the commercialization of LEQEMBI for the treatment of Alzheimer's disease and Sage on the commercialization of ZURZUVAE for the treatment of Postpartum Depression (PPD).
About the author

Emily J. Thompson
Emily J. Thompson, a Chartered Financial Analyst (CFA) with 12 years in investment research, graduated with honors from the Wharton School. Specializing in industrial and technology stocks, she provides in-depth analysis for Intellectia’s earnings and market brief reports.

FDA Approval: The FDA has approved a new high-dose regimen of Spinraza® (nusinersen) for the treatment of spinal muscular atrophy (SMA).
Treatment Impact: This approval is expected to enhance treatment options for patients suffering from SMA, a genetic disorder affecting muscle strength and movement.
See More
- Clinical Trial Success: Biogen's AMETHYST Phase 2/3 study of litifilimab demonstrated a statistically significant 11.8% reduction in skin disease activity compared to placebo at Week 16, indicating the potential for the drug to become the first innovative therapy for cutaneous lupus erythematosus in 70 years.
- Efficacy Assessment: The study revealed rapid and sustained improvements in skin disease activity over 24 weeks, with separation from placebo observed as early as Week 4, and a higher proportion of participants achieving CLASI-50 and CLASI-70 responses, underscoring the drug's effectiveness.
- Safety Profile: Over 24 weeks, adverse events occurred in 74.6% of litifilimab participants compared to 64.7% in the placebo group, with most events being mild to moderate; however, serious adverse events were reported in 6.8% of the litifilimab group, higher than the 2.9% in the placebo group.
- Market Reaction: Although Biogen's stock fell by 3.78% to $183.84 on March 27, it rebounded to $185.01 in after-hours trading, reflecting optimistic market sentiment regarding the drug's prospects.
See More
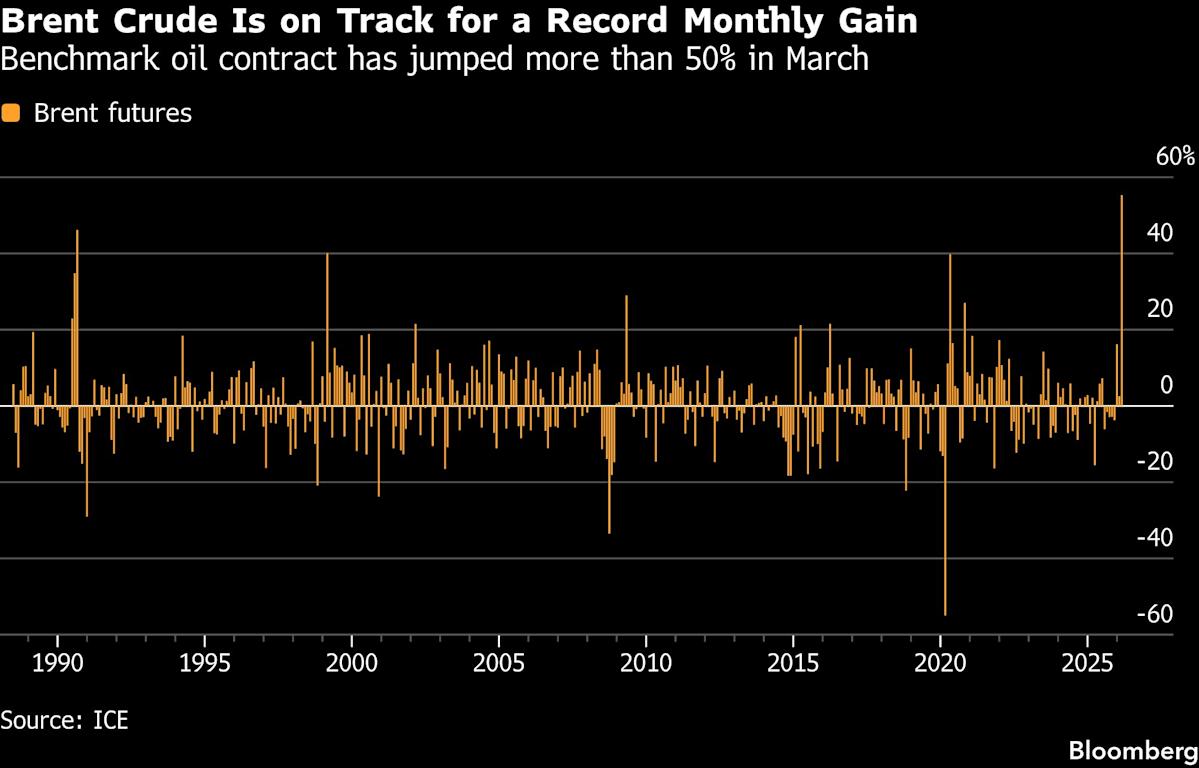
- Oil Price Surge Impacts Markets: The S&P 500 Index fell by 0.75%, the Dow Jones Industrial Average by 0.21%, and the Nasdaq 100 by 1.18% as the market reacts to the first oil settlement exceeding $100 since 2022, highlighting concerns over rising energy costs amid ongoing supply chain risks.
- Uncertain Fed Policy Outlook: Federal Reserve Chair Jerome Powell noted that while the central bank is monitoring the surge in energy prices, its tools to address supply-side shocks are limited, with markets pricing in only a 3% chance of a 25 bp rate hike at the April meeting, indicating investor uncertainty about future monetary policy.
- Escalating Middle East Tensions: US and Israeli forces continue military operations in Iran, with the Pentagon preparing for weeks of ground operations as approximately 3,500 sailors and Marines arrive in the region, potentially exacerbating global energy supply issues.
- Positive Bond Market Reaction: The 10-year Treasury yield fell to 4.330% as speculation grows that the ongoing conflict in the Middle East may lead to fuel shortages, offsetting inflation fears and supporting Treasury prices amid declining inflation expectations.
See More
- Mixed Stock Performance: The S&P 500 Index fell by 0.29%, while the Dow Jones Industrial Average rose by 0.26%, and the Nasdaq 100 Index decreased by 0.67%, indicating a complex market reaction to Federal Reserve policies and international tensions.
- Rising Oil Prices: Crude oil prices surged over 2% to a three-week high due to disruptions in oil and gas flows through the Strait of Hormuz caused by the Iran conflict, which could negatively impact global economic growth.
- Weak Manufacturing Activity: The Dallas Fed manufacturing activity index dropped from -0.4 to -0.2, falling short of the expected increase to 2.0, suggesting challenges in economic recovery that may influence future policy decisions.
- Escalating International Tensions: U.S. and Israeli military actions against Iran have intensified, with 3,500 sailors and Marines deployed to the Middle East, potentially leading to further volatility in energy markets and impacting global supply chains.
See More
- Market Gains: The S&P 500 Index rose by 0.58%, the Dow Jones Industrial Average increased by 0.91%, and the Nasdaq 100 Index climbed by 0.30%, reflecting a positive market sentiment amid expectations that the Fed may keep interest rates unchanged.
- Declining Bond Yields: The 10-year Treasury note yield fell by 10 basis points to 4.33%, driven by concerns that the ongoing war in the Middle East could lead to a fuel shortage, potentially hindering global economic growth and influencing the Fed's future rate decisions.
- Rising Oil Prices: Crude oil prices surged over 2% to a three-week high due to Iranian attacks on shipping, raising fears that the closure of the Strait of Hormuz could disrupt global oil and gas supplies, which may lead to significant price increases.
- Manufacturing Activity Decline: The Dallas Fed's manufacturing activity survey dropped by 0.4 to -0.2, falling short of the expected increase to 2.0, indicating signs of economic slowdown that could impact future investment decisions.
See More
- New Dosage Regimen: The US FDA has approved Biogen's new high-dose regimen for Spinraza, consisting of 50 mg/5 mL and 28 mg/5 mL doses, significantly enhancing treatment flexibility and efficacy.
- Accelerated Loading Phase: The new regimen allows individuals new to treatment to receive two 50 mg high-dose injections 14 days apart, followed by 28 mg maintenance doses every four months, thereby accelerating patient treatment progress.
- Transition for Low-Dose Patients: Patients switching from the low-dose regimen will follow their current dosing schedule every four months after a single high-dose loading phase, ensuring continuity and stability in their treatment.
- Clinical Study Support: The approval is supported by results from the phase 2/3 DEVOTE study, demonstrating significant advantages in treatment outcomes with the new dosing regimen, further solidifying Biogen's market position in spinal muscular atrophy.
See More