Edgewise Therapeutics Presents Long-Term Data on Sevasemten
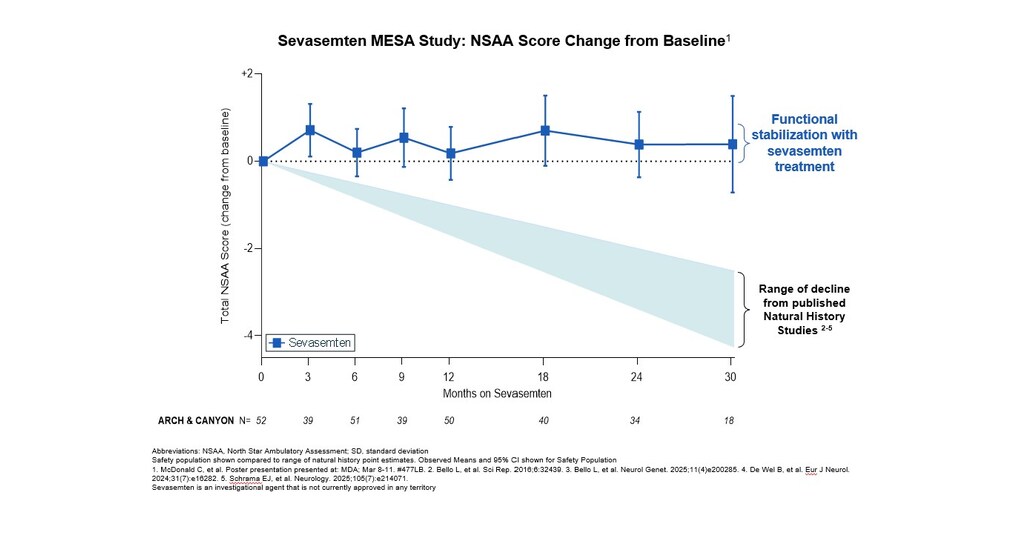
Edgewise Therapeutics presented long-term data from its MESA open-label extension study of sevasemten in Becker muscular dystrophy, a genetic disorder with no approved therapeutic interventions. The data show participants on sevasemten experienced stabilization of function over 3.5 years in marked contrast to the functional decline expected from Becker natural history data. Key findings reported after 12 months of ARCH and CANYON participation and further follow-ups in MESA: sustained stabilization over 3.5 years; divergence from Becker natural history. Sevasemten continued to show a favorable safety profile in MESA with long-term exposure for 3+ years. The company expects to announce top-line results of the sevasemten placebo-controlled pivotal cohort, GRAND CANYON, in 4Q. If data are positive, the company will advance sevasemten toward a marketing application to seek approval as the first targeted therapy for this underserved population.
Trade with 70% Backtested Accuracy
Analyst Views on EWTX
About EWTX
About the author

- Employee Incentive Plan: On March 31, 2026, Edgewise Therapeutics granted 72,000 stock options to four new non-executive employees, aimed at attracting talent and enhancing employee loyalty, aligning with the company's strategic goals under the 2024 Inducement Equity Incentive Plan.
- Option Exercise Price: Each stock option has an exercise price of $32.46 per share, equal to the closing price of Edgewise common stock on the grant date, ensuring alignment of employee interests with those of shareholders and promoting a performance-driven culture.
- Vesting Schedule: According to the inducement plan, 25% of the stock options will vest on the one-year anniversary of each employee's start date, with an additional 1/48th vesting monthly thereafter, designed to encourage long-term retention and continuous contribution to the company.
- Company Mission: Edgewise is dedicated to developing novel therapeutics for muscular diseases and serious cardiac conditions, committed to improving the quality of life for patients and their families, showcasing the company's deep expertise in biopharmaceuticals and social responsibility.
- Stock Option Grant: On March 31, 2026, Edgewise Therapeutics granted 72,000 stock options to four new non-executive employees, aimed at attracting talent and enhancing employee loyalty, in accordance with the company's 2024 Inducement Equity Incentive Plan.
- Exercise Price Setting: Each stock option has an exercise price of $32.46 per share, equal to the closing price of Edgewise common stock on the grant date, ensuring alignment of employee interests with those of shareholders.
- Vesting Arrangement: The vesting schedule for these stock options stipulates that 25% will vest on each employee's one-year anniversary, with an additional 1/48th vesting monthly thereafter, incentivizing continued service and contribution to the company.
- Compliance Assurance: This stock option grant complies with Nasdaq Listing Rule 5635(c)(4), demonstrating Edgewise's commitment to attracting and retaining key talent while enhancing its market credibility through compliance and transparency.
- Analyst Price Target Increase: Tessa Romero from JPMorgan raised Edgewise Therapeutics' price target from $34 to $45, resulting in a stock price increase of over 2%, significantly outperforming the S&P 500's 0.3% rise, indicating strong market confidence in the company.
- Drug Development Potential: The analyst highlighted that Edgewise has several investigational programs that could position it favorably in the biotech sector, particularly the renewed interest in EDG-7500, a drug targeting hypertrophic cardiomyopathy, which may drive future growth.
- Impact of Clinical Data Release: Last week, Edgewise published long-term data on sevasemten for muscular dystrophy, showing that Becker muscular dystrophy patients stabilized function with treatment, contrasting sharply with expected functional decline, thereby laying a solid foundation for the company's future market performance.
- Multiple Drug Development Advantage: Edgewise is not only developing one high-potential drug but also two additional drugs in the high-demand cardiovascular segment, making its diversified development strategy a standout feature in the biotech industry and attracting investor interest.
- Price Target Increase: Analyst Tessa Romero from JPMorgan raised Edgewise Therapeutics' price target from $34 to $45, an increase of $11, reflecting confidence in the company's pipeline drug potential, which is expected to attract more investor interest.
- Positive Market Reaction: Following the price target increase, Edgewise's stock rose over 2% on Tuesday, significantly outperforming the S&P 500's 0.3% gain, indicating a bullish sentiment among investors towards the biotech firm.
- Pipeline Drug Potential: The analyst highlighted Edgewise's EDG-7500 drug targeting the high-demand market for hypertrophic cardiomyopathy (HCM), which could propel the company to a leading position in the biotech sector.
- Clinical Data Support: Recent long-term data from Edgewise's muscular dystrophy treatment sevasemten showed stabilization of function in Becker-type patients, contrasting sharply with expected declines from natural history data, further bolstering market confidence in its products.
- Significant Functional Stability: The MESA study demonstrated that participants treated with sevasemten maintained functional stability over 3.5 years, contrasting sharply with the expected decline from Becker natural history studies, highlighting the drug's potential efficacy and safety.
- Clinical Trial Progress: Edgewise anticipates releasing pivotal GRAND CANYON data in Q4 2026, and if results are positive, will advance sevasemten towards a marketing application, potentially becoming the first targeted therapy for Becker, addressing a significant treatment gap.
- High Participant Engagement: With 99% of eligible participants from prior sevasemten studies opting to enroll in the MESA open-label extension, this high engagement reflects strong patient recognition and trust in the drug, enhancing the reliability of the study outcomes.
- Innovative Mechanism: Sevasemten, an orally administered fast skeletal myosin inhibitor, aims to reduce muscle damage, and its unique mechanism offers new hope for treating Becker and Duchenne muscular dystrophies, potentially transforming patients' quality of life.
- Stock Option Grant: On February 27, 2026, Edgewise Therapeutics granted 51,000 stock options to three new non-executive employees, aimed at attracting talent and strengthening the team, in accordance with the company's 2024 Inducement Equity Incentive Plan.
- Exercise Price Setting: Each inducement stock option has an exercise price of $30.44 per share, equal to the closing price of Edgewise common stock on the grant date, ensuring alignment of employee interests with those of shareholders.
- Vesting Arrangement: Under the plan, 25% of the stock options will vest on each employee's one-year anniversary, with an additional 1/48th vesting monthly thereafter, designed to incentivize long-term retention and contribution to the company's growth.
- Company Background: Edgewise is a biopharmaceutical company focused on muscle diseases, developing novel therapeutics for muscular dystrophies and serious cardiac conditions, dedicated to improving the quality of life for patients and their families.