Cytokinetics' Heart Drug Trial Success Boosts Stock
Written by Emily J. Thompson, Senior Investment Analyst
Updated: 1 hour ago
0mins
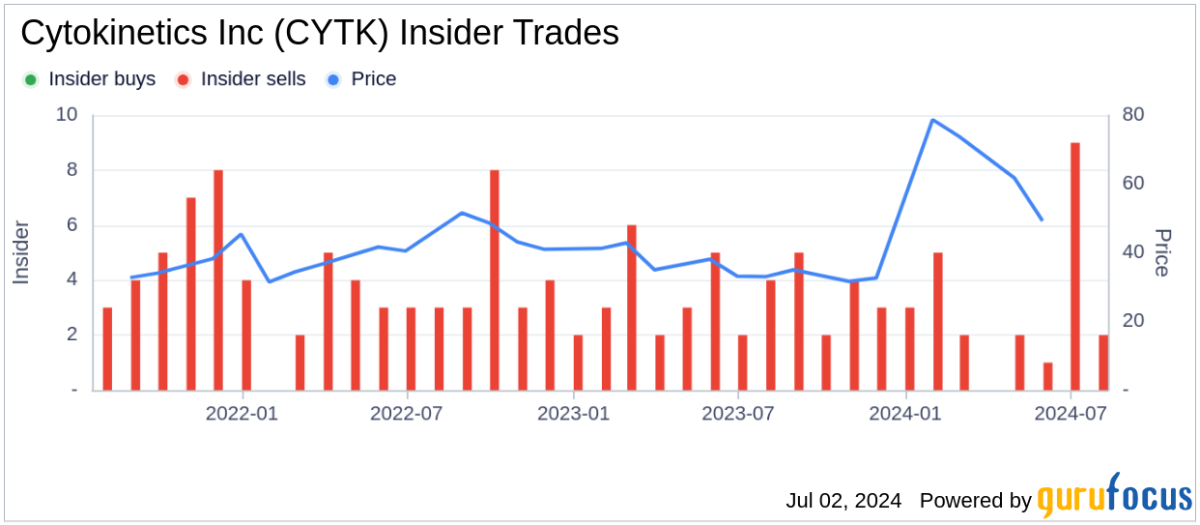
Should l Buy CYTK?
Source: stocktwits
- Significant Trial Results: Cytokinetics' Aficamten met primary endpoints in patients with symptomatic non-obstructive hypertrophic cardiomyopathy, showing substantial improvements in health-related quality of life and exercise capacity, indicating the drug's efficacy and market potential.
- Stock Price Surge: Following the trial results, Cytokinetics' shares soared over 16% on Tuesday, reaching their highest level in over two years, reflecting strong investor enthusiasm for the drug's prospects.
- Analyst Price Target Increase: Truist raised its price target for Cytokinetics from $92 to $106 while maintaining a 'Buy' rating, signaling increased market confidence in the company's growth potential.
- Competitive Landscape Analysis: Despite Cytokinetics' strong performance, analysts noted that Edgewise's EDG-7500 may be better positioned for treating hypertrophic cardiomyopathy, highlighting the competitive intensity and future market challenges in this therapeutic area.
Trade with 70% Backtested Accuracy
Stop guessing "Should I Buy CYTK?" and start using high-conviction signals backed by rigorous historical data.
Sign up today to access powerful investing tools and make smarter, data-driven decisions.
Analyst Views on CYTK
Wall Street analysts forecast CYTK stock price to rise
17 Analyst Rating
15 Buy
2 Hold
0 Sell
Strong Buy
Current: 66.050
Low
61.00
Averages
89.33
High
136.00
Current: 66.050
Low
61.00
Averages
89.33
High
136.00
About CYTK
Cytokinetics, Incorporated is a late-stage, specialty cardiovascular biopharmaceutical company focused on discovering, developing and commercializing muscle biology-directed drug candidates as potential treatments for debilitating diseases in which cardiac muscle performance is compromised. The Company is engaged in the commercialization of aficamten, a cardiac myosin inhibitor, and is being evaluated in additional clinical trials enrolling patients with obstructive and non-obstructive hypertrophic cardiomyopathy. The Company is also developing omecamtiv mecarbil, a cardiac myosin activator, in patients with heart failure with severely reduced ejection fraction (HFrEF); CK-586, a cardiac myosin inhibitor for the potential treatment of heart failure with preserved ejection fraction (HFpEF) and CK-089, a fast skeletal muscle troponin activator with potential therapeutic application to a specific type of muscular dystrophy and other conditions of impaired skeletal muscle function.
About the author

Emily J. Thompson
Emily J. Thompson, a Chartered Financial Analyst (CFA) with 12 years in investment research, graduated with honors from the Wharton School. Specializing in industrial and technology stocks, she provides in-depth analysis for Intellectia’s earnings and market brief reports.
- Results Announcement: Cytokinetics plans to report topline results from the ACACIA-HCM study on May 5, 2026, which aims to assess the impact of aficamten on cardiac endpoints in adults with non-obstructive hypertrophic cardiomyopathy, potentially providing critical data for future product development.
- Conference Call Details: Following the results announcement, the company will host a conference call at 8:00 AM Eastern Time to discuss the findings, with a simultaneous webcast available for investors and media, enhancing transparency and communication efficiency.
- Pipeline Overview: Cytokinetics focuses on cardiovascular biopharmaceuticals, leveraging over 25 years of innovation in muscle biology, with MYQORZO® approved in the U.S., Europe, and China for symptomatic obstructive hypertrophic cardiomyopathy, while also developing omecamtiv mecarbil and ulacamten, demonstrating ongoing commitment to heart disease treatment.
- Forward-Looking Statements: The company notes that all forward-looking statements are based on management's current expectations, with actual results potentially differing due to various risks and uncertainties, emphasizing the need for investors to carefully assess related risks.
See More
Cytokinetics Shares Surge: Cytokinetics' shares increased by 23.7% in premarket trading following positive developments related to a heart disease drug.
Focus on Heart Disease Treatment: The company's latest study aims to meet significant goals in the treatment of late-stage heart disease, highlighting its commitment to advancing medical solutions.
See More
- Clinical Trial Success: Cytokinetics' ACACIA-HCM trial successfully met dual primary endpoints, with KCCQ-CSS score improving by 11.4 points and pVO2 increasing by 0.64 mL/kg/min, indicating aficamten's efficacy in patients with non-obstructive hypertrophic cardiomyopathy, potentially transforming treatment paradigms in this area.
- Significant Patient Benefits: The trial results demonstrate that aficamten significantly improves exercise capacity and symptom burden, particularly with sustained improvements in KCCQ scores, highlighting the drug's potential value in clinical applications.
- Good Safety Profile: The safety of aficamten was comparable to placebo, with 88.4% of patients completing planned dosing and no new safety signals identified, enhancing confidence in the drug's profile.
- Future Outlook: Cytokinetics plans to present ACACIA-HCM results at an upcoming medical meeting and discuss with the FDA and other regulatory authorities, indicating a promising path for aficamten's potential approval in treating non-obstructive hypertrophic cardiomyopathy.
See More

Positive Trial Results: Cytokinetics announced positive topline results from a Phase 3 clinical trial of aficamten in patients with non-obstructive hypertrophic cardiomyopathy.
Patient Focus: The trial specifically targeted patients suffering from this condition, highlighting the potential benefits of aficamten for improving their health outcomes.
See More
- Strong Earnings Performance: Rockwell Automation reported a fiscal Q2 earnings per share of $3.30, exceeding analyst expectations of $2.88, with revenue of $2.24 billion surpassing the forecast of $2.16 billion, indicating robust market demand and operational efficiency.
- Upgraded Outlook: The company raised its fiscal 2026 profit outlook, reflecting confidence in future performance, which is likely to further boost investor sentiment and stock price.
- Competitor Struggles: Huntington Ingalls saw an 11% drop in shares despite free cash flow projections of $500 million to $600 million, falling short of the consensus estimate of $569.7 million, highlighting increasing competitive pressures in the industry.
- Positive Market Reaction: Other companies like DuPont and Waters Corp also saw stock price increases due to better-than-expected earnings, indicating a recovering market confidence in the manufacturing and technology sectors.
See More
- Significant Trial Results: Cytokinetics' Aficamten met primary endpoints in patients with symptomatic non-obstructive hypertrophic cardiomyopathy, showing substantial improvements in health-related quality of life and exercise capacity, indicating the drug's efficacy and market potential.
- Stock Price Surge: Following the trial results, Cytokinetics' shares soared over 16% on Tuesday, reaching their highest level in over two years, reflecting strong investor enthusiasm for the drug's prospects.
- Analyst Price Target Increase: Truist raised its price target for Cytokinetics from $92 to $106 while maintaining a 'Buy' rating, signaling increased market confidence in the company's growth potential.
- Competitive Landscape Analysis: Despite Cytokinetics' strong performance, analysts noted that Edgewise's EDG-7500 may be better positioned for treating hypertrophic cardiomyopathy, highlighting the competitive intensity and future market challenges in this therapeutic area.
See More







