Apogee Therapeutics Presents New Data on Zumilokibart in Phase 2 APEX Trial
- Clinical Trial Update: Apogee Therapeutics is set to present 52-week results from Part A of its Phase 2 APEX trial for Zumilokibart (APG777) on March 23, 2026, during a company webcast, marking a significant milestone for its anti-IL-13 antibody program.
- Dosing Strategy Innovation: Zumilokibart is engineered for extended half-life and infrequent dosing, with potential administration every three or six months, which Apogee believes could establish a new standard in the atopic dermatitis market.
- Trial Design: The APEX trial consists of two complementary parts, with Part A being a 52-week maintenance phase aimed at evaluating long-term safety, durability of response, and the ability to maintain skin clearance over a full year, and today's readout will clarify the effectiveness of its extended-interval dosing strategy.
- Future Development Plans: Apogee plans to advance Zumilokibart into a Phase 3 trial in the second half of 2026, based on results from both Part A and Part B, along with upcoming regulatory discussions to refine dose selection for late-stage development.
Trade with 70% Backtested Accuracy
Analyst Views on APGE
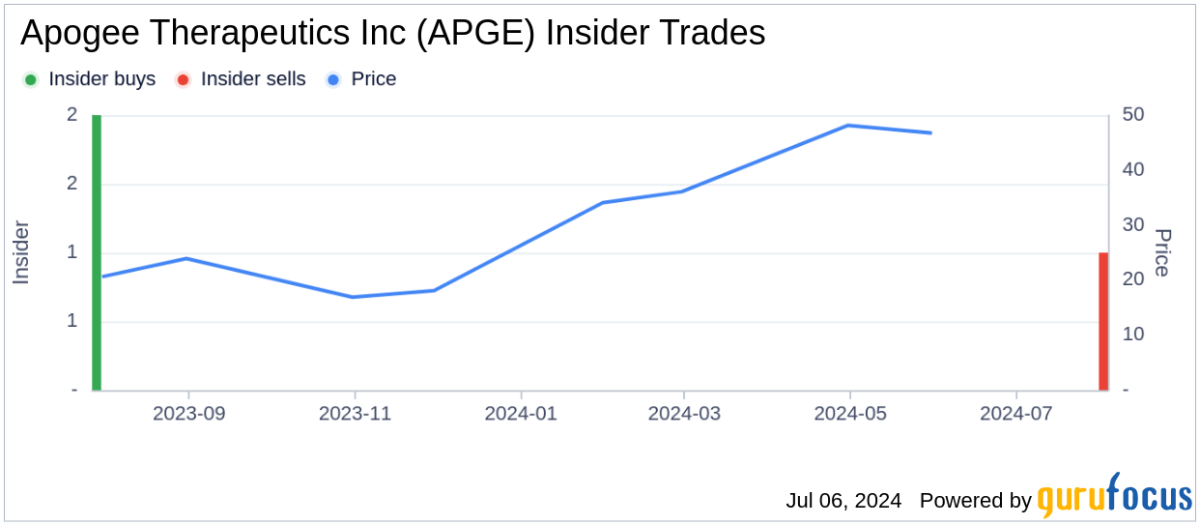
About APGE
About the author

Company Performance: A company named Apogee has reported a 16.6% increase in its pre-market shares.
Drug Development Update: The rise in shares follows the announcement of positive results from a mid-stage trial for a drug targeting eczema.

Therapeutics Overview: The article discusses the therapeutic agent ZUMILOKIBART, highlighting its safety profile and efficacy.
Safety Profile: ZUMILOKIBART is noted to have a safety profile consistent with other agents in its class, indicating it is well-tolerated.

Therapeutics in Deepening Response: The article discusses the advancements in therapeutic approaches aimed at enhancing response mechanisms in various medical conditions.
Observations Across Lesional and Itch Points: It highlights the observations made in clinical settings regarding lesional areas and itch points, emphasizing their significance in treatment outcomes.
Dosing Strategies: The content outlines the dosing strategies employed in studies, specifically focusing on 3-month and 6-month dosing regimens.
Implications for Future Research: The findings suggest potential implications for future research directions in therapeutic interventions and patient response evaluations.
- Clinical Trial Update: Apogee Therapeutics is set to present 52-week results from Part A of its Phase 2 APEX trial for Zumilokibart (APG777) on March 23, 2026, during a company webcast, marking a significant milestone for its anti-IL-13 antibody program.
- Dosing Strategy Innovation: Zumilokibart is engineered for extended half-life and infrequent dosing, with potential administration every three or six months, which Apogee believes could establish a new standard in the atopic dermatitis market.
- Trial Design: The APEX trial consists of two complementary parts, with Part A being a 52-week maintenance phase aimed at evaluating long-term safety, durability of response, and the ability to maintain skin clearance over a full year, and today's readout will clarify the effectiveness of its extended-interval dosing strategy.
- Future Development Plans: Apogee plans to advance Zumilokibart into a Phase 3 trial in the second half of 2026, based on results from both Part A and Part B, along with upcoming regulatory discussions to refine dose selection for late-stage development.
- Data Release Announcement: Apogee Therapeutics is set to report 52-week data from its Phase 2 APEX trial on March 23, 2026, marking a significant advancement in its efforts to address conditions like atopic dermatitis, which could influence market expectations for its products.
- Webcast Details: The company will host a live webcast at 8:00 a.m. ET on the same day to discuss the trial results, which is expected to attract attention from investors and industry experts, thereby enhancing the company's visibility in the biotechnology sector.
- Product Development Outlook: Zumilokibart, Apogee's leading development program, targets unmet needs in atopic dermatitis and other indications; positive results could help the company secure a competitive edge in the crowded immunology market.
- Market Potential Analysis: Apogee's pipeline includes treatments for asthma and chronic obstructive pulmonary disease, and achieving best-in-class efficacy and dosing could present significant market opportunities, further driving growth in the biopharmaceutical industry.
- Clinical Trial Results: Apogee Therapeutics announced positive 52-week maintenance data from part A of the Phase 2 APEX trial for zumilokibart (APG777) in moderate-to-severe atopic dermatitis patients, demonstrating significant durability of response with both 3- and 6-month maintenance dosing regimens, positioning it as a potential best-in-class anti-IL-13 antibody.
- Efficacy Improvement: The results indicated a deepening of response across all lesional and itch endpoints, supporting zumilokibart's differentiated profile, particularly its significantly less frequent dosing compared to current standard of care, which could enhance patient treatment experiences.
- Future Trial Plans: The APEX part B 16-week induction results are expected to be released in Q2 2026, providing support for the anticipated initiation of Phase 3 trials for zumilokibart in moderate-to-severe atopic dermatitis in the second half of 2026, further validating its therapeutic potential.
- Positive Market Reaction: Apogee Therapeutics shares rose 18% to $78.00 in early trading, reflecting strong market interest and confidence in its new therapy, which could transform the treatment landscape for atopic dermatitis.








