Actinium Pharmaceuticals Presents Actimab-A Anti-Leukemic Activity Data at AACR Meeting
Actinium Pharmaceuticals highlighted data presented at the American Association for Cancer Research, or AACR, Annual Meeting supporting transcriptional reprogramming as a central mechanism driving the mutation-agnostic anti-leukemic activity of Actimab-A, or lintuzumab-Ac225, in acute myeloid leukemia, or AML. Preclinical translational data demonstrated that lintuzumab-Ac225 delivers potent cytotoxic activity across AML models harboring common mutations, including FLT3, NPM1, KMT2A, and TP53, as well as in primary patient samples. Importantly, combining Actimab-A with standard-of-care therapies - the menin inhibitor revumenib, the FLT3 inhibitor gilteritinib, and the hypomethylating agent azacitidine - resulted in enhanced leukemic cell killing in vivo across all tested models, independent of mutation status. These results support a combination-driven clinical strategy aimed at improving depth and durability of response. The findings provide the mechanistic foundation for Actimab-A's observed clinical activity and, together with the manageable safety profile demonstrated across prior Actimab-A trials in more than 150 AML patients, reinforce its suitability as a combination backbone across multiple treatment settings. Actimab-A is Actinium's lead clinical radiotherapy delivering Actinium-225, a potent alpha-emitter radioisotope payload that produces lethal double-strand DNA breaks to kill CD33-expressing AML cells. In the relapsed/refractory AML setting Actimab-A in combination with the intensive chemotherapy regimen CLAG-M produced an 83% overall response rate and 75% MRD-negativity in a Phase 1 trial which forms the basis of a Phase 2/3 registrational study for which Actinium has FDA alignment and is seeking a development partner. Actimab-A is also being studied in newly diagnosed patients via the ongoing NCI-sponsored frontline triplet trial of Actimab-A with venetoclax and ASTX-727; and has shown promise in post-remission and MRD-directed settings; as well as myelodysplastic syndrome and other CD33-expressing myeloid malignancies. Combination treatment produced consistent pathway-level changes compared with monotherapy. Gene set enrichment analyses showed enhanced myeloid differentiation signatures with the addition of Actimab-A (lintuzumab-Ac225) to revumenib, gilteritinib, and azacitidine. Together, these findings show that Actimab-A combinations don't just add cytotoxicity - they reprogram AML cells from proliferation toward differentiation and apoptosis, providing the mechanistic basis for deeper, more durable MRD-negative responses and reinforcing Actimab-A's role as a universal combination backbone across AML.
Trade with 70% Backtested Accuracy
Analyst Views on ATNM

No data
About ATNM
About the author

- Optimistic Outlook for SLS009: Sellas is set to present preclinical AML data on SLS009 at the AACR meeting, demonstrating significant increases in cancer cell death in models with ASXL1 and TP53 mutations, which could attract investor attention and drive stock price appreciation.
- ATNM Data Highlights: Actinium will showcase new data from its Actinium-225 radiotherapy platform at AACR, particularly regarding ATNM-400 and Actimab-A across leukemia and solid tumor programs, potentially boosting market confidence in its treatment options.
- Analyst Target Discrepancies: According to consensus estimates from Koyfin, ATNM has a 12-month average analyst price target of $5.75, implying a 342% upside, while SLS's target is $8.67, suggesting about 66% upside, indicating a higher appeal for ATNM among investors.
- Retail Sentiment Comparison: On Stocktwits, retail sentiment for ATNM is deemed 'extremely bullish', while SLS appears 'bearish', which may influence investor decisions and market performance.
- Data Presentation: Actinium Pharmaceuticals will present new data for ATNM-400 and Actimab-A at the 2026 AACR Annual Meeting, highlighting their broad efficacy across multiple solid tumor models, which could potentially redefine current treatment standards.
- ATNM-400 Potential: As a first-in-class Ac-225 radioconjugate, ATNM-400 demonstrates the ability to combat various tumors, particularly those resistant to existing targeted therapies, positioning it as a leading treatment option for large solid tumor indications.
- Actimab-A Mechanism Innovation: Actimab-A's newly identified mechanism enhances responses to standard acute myeloid leukemia (AML) therapies through transcriptional reprogramming, showcasing its mutation-agnostic efficacy and offering new treatment options for AML patients.
- Future Outlook: Multiple data catalysts for ATNM-400 and Actimab-A are expected in 2026, with Actinium's strategy focused on continuous innovation and a diversified product pipeline, which is anticipated to drive significant value opportunities for the company.
- Investor Attention: As the earnings season unfolds, mid to low market capitalization healthcare stocks are drawing investor attention due to their strong earnings momentum, indicating growing market confidence in this sector.
- Analyst Expectations: The EPS Revision Grade reflects the trend in analyst earnings estimates, with A+ ratings indicating optimistic projections for future performance, potentially driving stock prices higher.
- List of A+ Rated Stocks: Currently, companies such as Aldeyra Therapeutics, Altimmune, Annovis Bio, and Assertio Holdings have received A+ EPS Revision Grades, showcasing their strong performance in the eyes of analysts.
- Market Strategy Impact: These A+ rated healthcare stocks are likely to attract more investor interest, potentially triggering positive sentiment towards the healthcare sector as a whole, thereby enhancing the performance of related ETFs.
- FibroBiologics Outperformance: FibroBiologics, Inc. (FBLG) surged 7.68% in after-hours trading to close at $0.41, indicating speculative interest or technical momentum despite no specific news.
- Nyxoah Earnings Boost Confidence: Nyxoah SA (NYXH) advanced 3.94% to $5.28 after reporting preliminary Q4 and full-year 2025 results, with guidance for Q1 2026 enhancing investor confidence in its growth trajectory.
- Fortress Biotech's Continued Volatility: Fortress Biotech, Inc. (FBIO) climbed 6.90% to $4.49 in after-hours trading, continuing a trend of volatility without any fresh news impacting the stock.
- Revvity Collaboration Drives Growth: Revvity, Inc. (RVTY) posted a 4.92% gain to close at $109.00, as investors digest the January announcement of a collaboration with Eli Lilly to expand access to predictive models via the Revvity Signals platform.
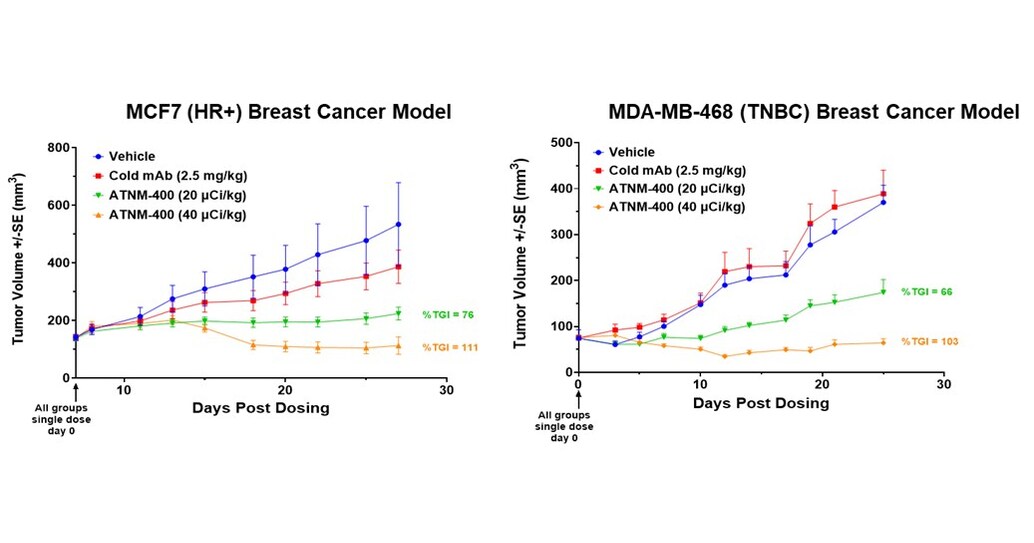
- Strong Anti-Tumor Activity: ATNM-400 demonstrated significant tumor growth inhibition across various breast cancer models, particularly in hormone receptor-positive and triple-negative cases, with all treatment regimens well tolerated and no significant body weight changes observed, indicating its potential clinical value.
- Effectiveness Against Resistant Tumors: The drug induced irreversible DNA damage in standard treatment-resistant breast cancer models, showcasing its unique advantage in addressing resistant tumors and potentially offering new treatment options for patients.
- Broad Applicability: ATNM-400 not only shows strong efficacy in breast cancer but also exhibits promising anti-tumor activity in prostate cancer and non-small cell lung cancer, supporting its potential as a targeted radiotherapy for multiple solid tumors.
- Market Demand Alignment: With an estimated increase to 250,000 women living with metastatic breast cancer by 2030, the development of ATNM-400 aligns perfectly with this growing market need, potentially providing new hope for patients with hard-to-treat breast cancer.
- Potent Anti-Tumor Activity: ATNM-400 demonstrated significant tumor growth inhibition across various breast cancer models, particularly in hormone receptor-positive and triple-negative types, with all treatment regimens well tolerated and no significant weight changes observed, indicating its potential for clinical application.
- Efficacy in Resistant Models: In trastuzumab- and tamoxifen-resistant breast cancer cells, ATNM-400 exhibited enhanced cytotoxicity, and combining it with standard treatments resulted in greater cytotoxicity and tumor regression, showcasing its promise as a combination therapy.
- Mechanistic Evidence of DNA Damage: Treatment with ATNM-400 led to irreversible double-strand DNA breaks in breast cancer cells, activating AKT phosphorylation, which indicates its effective mechanism in resistant models and may provide new options for treating hard-to-treat breast cancer.
- Broad Applicability: Beyond breast cancer, ATNM-400 has shown potential efficacy in prostate cancer and non-small cell lung cancer, supporting its development as a multi-indication targeted radiotherapy to meet clinical needs.









