U.S. Stock Futures Rise, Airline Stocks Benefit
Stock futures are higher this morning. Stocks came into the session under heavy pressure after global equities slid to four-month lows, reacting to an 80% surge in oil prices this year and the growing risk of supply disruptions through the Strait of Hormuz.However, oil prices are pulling back, the major averages are jumping, and shares of airlines and cruiselines are up in pre-market trading in the wake of President Trump's message that the U.S. and Iran had "very good and productive conversations" regarding a "complete resolution of hostilities".In pre-market trading, S&P 500 futures rose 1.93%, Nasdaq futures rose 2.04% and Dow futures rose 2.11%.Check out this morning's top movers from around Wall Street, compiled by The Fly.HIGHER -Apogee Therapeuticsup 16% after announcing a 52-week maintenance data from Part A of the Phase 2 APEX clinical trial of zumilokibartFlutter Entertainmentand DraftKingsup 9% and 6%, respectively, after The Wall Street JournalU.S. senators have introduced legislation that will prohibit entities regulated by the Commodity Futures Trading Commission from listing contracts related to sporting eventsSynopsysup 3% after WSJ reports that activist investor Elliot Management has built a multibillion-dollar investment in Synopsys and plans to push the company to make more money from its software and servicesSolo Brandsup 1o% after introducing FY26 guidance ahead of its investor dayDOWN AFTER OIL PRICE DECLINE-Occidentaldown 4%BPdown 3%ConocoPhillipsdown 2%Exxon Mobildown 2%TotalEnergiesdown 1%EOG Resourcesdown 1%Chevrondown 1%Shelldown 1%LOWER -Gossamer Biodown 5% after Cantor Fitzgerald downgraded the stock to Neutral without a price targetN-abledown 5% after William Blair double downgraded shares to Underperform from Outperform without a price target
Trade with 70% Backtested Accuracy
Analyst Views on APGE
About APGE
About the author

- Price Range Analysis: The XBI ETF's 52-week low is $66.66 per share, with a high of $132.09, and a recent trading price of $119.75, indicating stability within its volatility range that may attract investor interest.
- Technical Analysis Tool: Comparing the current share price to the 200-day moving average can provide valuable insights for investors, aiding in market trend assessment and timing of investments.
- ETF Trading Mechanism: ETFs trade like stocks, where investors buy and sell 'units' that can be created or destroyed based on demand, impacting the liquidity and market performance of the ETF.
- Inflows and Outflows Monitoring: Weekly monitoring of changes in ETF shares outstanding focuses on significant inflows (new units created) or outflows (old units destroyed), which will affect the underlying holdings of the ETF and influence market dynamics.
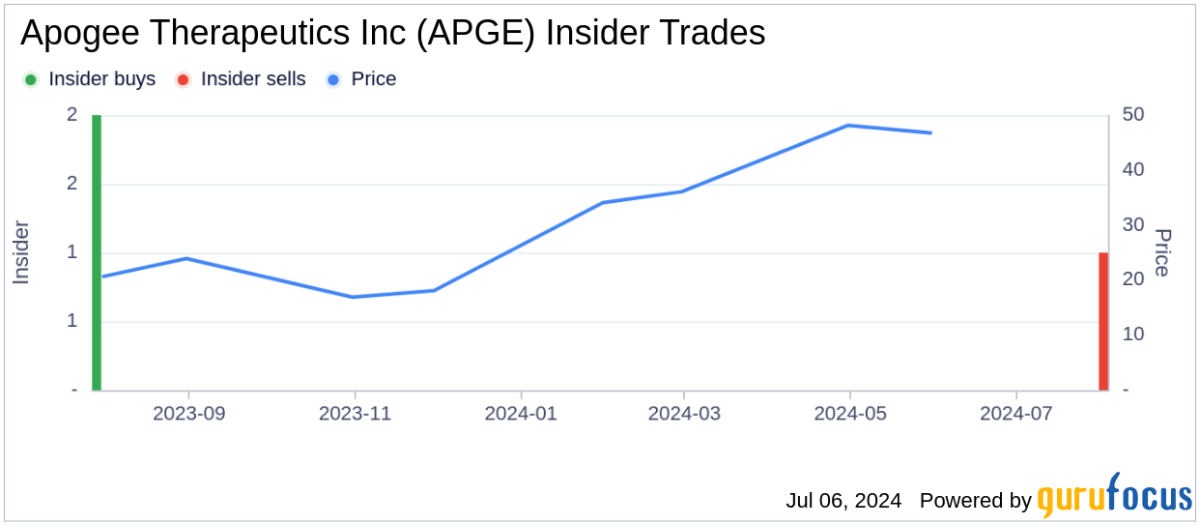
- Executive Stock Sale: Apogee Therapeutics CEO Michael Henderson sold 20,000 shares of common stock on March 11, 2026, for a total value of $1.5 million, retaining approximately 1.15 million shares valued at around $86.11 million post-transaction, indicating confidence in the company's future prospects.
- Transaction Scale Analysis: The sale of 20,000 shares aligns with Henderson's recent average sale size, representing 1.71% of his direct holdings, consistent with his historical trading pattern, which reflects stability and consistency in the market.
- Company Financial Status: Apogee Therapeutics has a current market capitalization of $5.5 billion, and despite a trailing twelve-month net income of -$255.84 million, its stock price has surged by 98% over the past year, demonstrating strong market confidence in its biologic therapeutic products.
- Future Development Outlook: The company recently completed a $350 million equity offering expected to close on Thursday, which will strengthen its financial position and support ongoing clinical development, particularly in the upcoming Phase 3 trials, making it crucial for investors to focus on the forthcoming positive data releases.
- Offering Size: Apogee Therapeutics has announced a public offering of 5,000,000 shares at a price of $70.00 per share, with expected gross proceeds of approximately $350 million, which will significantly bolster the company's funding for R&D and market expansion.
- Underwriter Arrangement: The offering is managed by Jefferies, TD Cowen, Stifel, and Guggenheim Securities, reflecting strong market confidence in Apogee and potentially enhancing its reputation within the biotechnology sector.
- Additional Share Option: Apogee has granted underwriters a 30-day option to purchase an additional 750,000 shares, providing flexibility that may further enhance the company's capital-raising capabilities to meet future market demands.
- Market Outlook: With a focus on treating major immunological diseases like atopic dermatitis, Apogee's innovative biologics are expected to attract increased investor interest, thereby boosting the company's competitiveness in the rapidly growing biotechnology market.
- Upsized Offering: Apogee Therapeutics has announced an upsized public offering of 5 million shares at $70.00 per share, with total gross proceeds expected to reach approximately $350 million, reflecting strong market demand compared to the previously announced $300 million target.
- Underwriter Option: The offering includes an option for underwriters to purchase up to 750,000 additional shares within 30 days at the same price, enhancing the company's financial flexibility and potential capital raise capabilities.
- Closing Timeline: The offering is set to close on March 26, 2026, marking a proactive approach by Apogee in the capital markets to secure funding for future research and market expansion initiatives.
- Positive Market Reaction: Following the announcement of the offering, Apogee Therapeutics' stock price rose due to positive developments in its eczema treatment, indicating investor confidence in the company's future potential.
- Offering Size: Apogee Therapeutics announced the pricing of 5,000,000 shares at $70.00 each, expecting gross proceeds of approximately $350 million, which will significantly support the company's R&D and market expansion efforts.
- Underwriter Arrangement: Jefferies, TD Cowen, Stifel, and Guggenheim Securities are acting as joint book-running managers for the offering, ensuring professional management and market credibility, thereby enhancing investor confidence.
- Additional Share Option: Apogee granted underwriters a 30-day option to purchase an additional 750,000 shares, a flexible arrangement that could further increase total proceeds and enhance the company's financial flexibility.
- Market Outlook: The successful offering provides funding for Apogee's innovative biologics targeting conditions like atopic dermatitis and asthma, expected to boost its competitiveness in the inflammatory and immunology markets, helping the company achieve best-in-class efficacy and dosing objectives.
- Funding Size: Apogee Therapeutics has announced a public offering of $300 million in common stock, which is intended to fund clinical trials and commercial readiness activities for its antibody programs, thereby enhancing its competitive edge in the biopharmaceutical sector.
- Stock Price Reaction: Following the announcement, APGE's stock rose by 2.04% to $80.86, reflecting market confidence in the company's future, especially after the positive clinical data release for its lead drug.
- Underwriter Arrangement: Apogee plans to grant underwriters a 30-day option to purchase up to an additional $45 million in common stock, a strategy that will provide the company with greater financial flexibility to support its R&D efforts.
- Clinical Progress: The company's lead candidate, Zumilokibart, has shown positive 52-week Phase 2 data for atopic dermatitis, further solidifying its market position in treating inflammatory and immunological diseases.