REGENXBIO Faces Securities Litigation Investigation
Written by Emily J. Thompson, Senior Investment Analyst
Updated: 4 hours ago
0mins
Should l Buy RGNX?
Source: Globenewswire
- Legal Investigation Initiated: Faruq & Faruq LLP is investigating potential securities fraud claims against REGENXBIO related to securities transactions from February 9, 2022, to January 27, 2026, with a deadline for investors to apply as lead plaintiffs by April 14, 2026, highlighting the urgency and significance of the case.
- FDA Clinical Hold: On January 28, 2026, REGENXBIO announced that its gene therapies RGX-111 and RGX-121 faced clinical holds from the FDA due to a tumor case in a participant, leading to a 17.9% drop in stock price on the same day, significantly impacting investor confidence.
- Investor Loss Warning: The company's failure to disclose material adverse facts regarding the efficacy and safety of RGX-111 may result in substantial financial losses for investors, prompting Faruq & Faruq to encourage affected investors to contact them directly to discuss legal options, demonstrating a commitment to investor rights.
- Class Action Opportunity: Any eligible investor can apply to serve as lead plaintiff through counsel or choose to remain absent, indicating the collective nature of the lawsuit and its potential impact on investors, emphasizing the complexity of legal proceedings and the rights of investors.
Trade with 70% Backtested Accuracy
Stop guessing "Should I Buy RGNX?" and start using high-conviction signals backed by rigorous historical data.
Sign up today to access powerful investing tools and make smarter, data-driven decisions.
Analyst Views on RGNX
Wall Street analysts forecast RGNX stock price to rise
7 Analyst Rating
7 Buy
0 Hold
0 Sell
Strong Buy
Current: 9.380
Low
19.00
Averages
29.71
High
45.00
Current: 9.380
Low
19.00
Averages
29.71
High
45.00
About RGNX
REGENXBIO Inc. is a clinical-stage biotechnology company seeking to improve lives through the curative potential of gene therapy. The Company has developed a pipeline of gene therapy programs using its proprietary adeno-associated virus (AAV) gene therapy delivery platform (NAV Technology Platform) to address genetic diseases. It is focused on its internal development pipeline in three areas: retinal, neuromuscular, and neurodegenerative diseases. Its investigational AAV therapeutics include ABBV-RGX-314, RGX-202, RGX-121, and RGX-111. It is developing ABBV-RGX-314 in collaboration with AbbVie to treat large patient populations impacted by wet age-related macular degeneration, diabetic retinopathy (DR) and other chronic retinal diseases characterized by loss of vision. It is developing RGX-202 to treat Duchenne muscular dystrophy (Duchenne). The Company is developing RGX-121 to treat Mucopolysaccharidosis type II (MPS II), a progressive, neurodegenerative lysosomal storage disorder.
About the author

Emily J. Thompson
Emily J. Thompson, a Chartered Financial Analyst (CFA) with 12 years in investment research, graduated with honors from the Wharton School. Specializing in industrial and technology stocks, she provides in-depth analysis for Intellectia’s earnings and market brief reports.
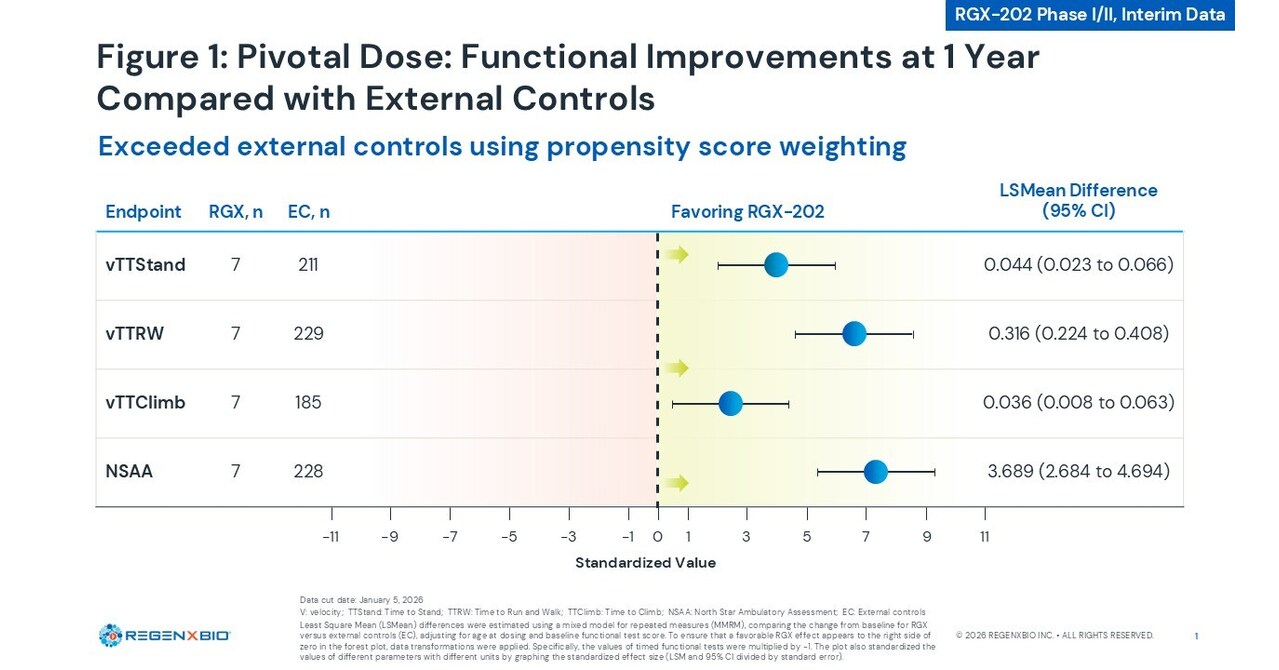
- Clinical Trial Progress: REGENXBIO has announced positive interim data from the Phase I/II AFFINITY DUCHENNE trial of RGX-202, indicating significant improvements in functionality, safety, and biomarkers, positioning it as a potential best-in-class gene therapy for Duchenne muscular dystrophy.
- Expert Data Presentation: The trial's principal investigator, Dr. Carolina Tesi-Rocha, Clinical Professor of Neurology at Stanford University School of Medicine, presented these findings at the Muscular Dystrophy Association (MDA) Clinical and Scientific Conference, enhancing the therapy's academic recognition and industry visibility.
- Cardiac MRI Assessments: The new data includes cardiac MRI evaluations, suggesting RGX-202's potential in improving cardiac function, which is crucial for the overall health of Duchenne muscular dystrophy patients and may influence future treatment protocols.
- Optimistic Market Outlook: With positive clinical feedback, REGENXBIO is poised to carve out a niche in the competitive gene therapy market, attracting investor interest and potentially driving up the company's stock price.
See More
- Class Action Filed: Pomerantz LLP has initiated a class action lawsuit against Regenxbio, alleging securities fraud and other unlawful business practices by the company and certain officers, with investors needing to apply as Lead Plaintiff by April 14, 2026, indicating significant legal risks that could impact the company's reputation and stock price.
- FDA Clinical Hold: On January 28, 2026, Regenxbio announced that the FDA placed a clinical hold on its gene therapies RGX-111 and RGX-121 due to a neoplasm case found in trials, leading to a 17.9% drop in stock price on the same day, reflecting the direct impact of regulatory risks on the company's operations.
- Significant Stock Decline: Following the FDA announcement, Regenxbio's stock price fell from $13.41 to $11.01, a decrease of $2.40, highlighting market concerns regarding the company's future prospects, which may affect investor confidence and its ability to raise funds.
- Legal Implications: Pomerantz LLP is a prominent law firm specializing in class action litigation related to securities fraud and corporate misconduct, and if the lawsuit succeeds, Regenxbio could face substantial damages, further exacerbating its financial pressures.
See More
- Legal Investigation Initiated: Faruq & Faruq LLP is investigating potential securities fraud claims against REGENXBIO related to securities transactions from February 9, 2022, to January 27, 2026, with a deadline for investors to apply as lead plaintiffs by April 14, 2026, highlighting the urgency and significance of the case.
- FDA Clinical Hold: On January 28, 2026, REGENXBIO announced that its gene therapies RGX-111 and RGX-121 faced clinical holds from the FDA due to a tumor case in a participant, leading to a 17.9% drop in stock price on the same day, significantly impacting investor confidence.
- Investor Loss Warning: The company's failure to disclose material adverse facts regarding the efficacy and safety of RGX-111 may result in substantial financial losses for investors, prompting Faruq & Faruq to encourage affected investors to contact them directly to discuss legal options, demonstrating a commitment to investor rights.
- Class Action Opportunity: Any eligible investor can apply to serve as lead plaintiff through counsel or choose to remain absent, indicating the collective nature of the lawsuit and its potential impact on investors, emphasizing the complexity of legal proceedings and the rights of investors.
See More
- Class Action Notice: Rosen Law Firm reminds investors who purchased REGENXBIO (NASDAQ: RGNX) securities between February 9, 2022, and January 27, 2026, to apply as lead plaintiffs by April 14, 2026, to potentially receive compensation without any out-of-pocket costs.
- Lawsuit Background: The lawsuit alleges that defendants provided misleading information regarding the development of RGX-111 gene therapy, claiming positive biomarker and safety data while concealing material adverse facts about the therapy's efficacy and safety, resulting in investor losses when the truth emerged.
- Law Firm Credentials: Rosen Law Firm specializes in securities class actions and has recovered over $438 million for investors in 2019 alone, achieving the largest securities class action settlement against a Chinese company in 2017, showcasing its strong track record and expertise in this field.
- Participation Instructions: Investors can visit Rosen Law Firm's website or call toll-free at 866-767-3653 for more information, emphasizing the importance of selecting qualified legal counsel, especially since the class has not yet been certified, leaving investors to choose their representation wisely.
See More
- Positive Clinical Data: REGENXBIO announced that interim data from its early-to-mid stage trial of the DMD gene therapy RGX-202 shows evidence of positively changing disease trajectory in some patients, indicating potential new hope for DMD patients.
- Favorable Safety Profile: The company noted that RGX-202 exhibited no serious adverse events and no evidence of liver injury, suggesting that a proactive short-course immune modulation regimen combined with high product purity may contribute to its favorable safety profile, enhancing market confidence.
- Application Timeline: REGENXBIO expects to share topline data from the trial in early Q2 2024 and plans to request a meeting with the FDA in mid-2026, indicating proactive steps in advancing the approval process.
- Market Sentiment: Despite RGNX stock gaining nearly 54% over the past 12 months, sentiment on Stocktwits shifted from 'extremely bullish' to 'bullish', reflecting fluctuations in market attention towards the therapy.
See More
- Clinical Trial Results: Regenxbio's interim data from the RGX-202 trial for Duchenne muscular dystrophy shows participants improved an average of 4.9 points on the North Star Ambulatory Assessment (NSAA), indicating potential efficacy, yet the stock price dropped, reflecting market concerns about long-term outcomes.
- Biomarker Data: New data reveals consistent high expression of RGX-202 microdystrophin in 13 patients, with one 3.6-year-old patient showing a 51.2% expression level at week 12, suggesting biological effectiveness of the therapy.
- FDA Review Dynamics: The FDA issued a Complete Response Letter for RGX-121's Biologics License Application in February, indicating regulatory challenges that may impact investor confidence and stock performance.
- Technical Analysis Status: Regenxbio shares are currently trading 7.5% above their 20-day simple moving average (SMA) but 22.6% below the 100-day SMA, indicating a lack of long-term momentum, necessitating close monitoring of the key support level at $7.50.
See More










