MLYS.O Falls Below 20-Day SMA Amid Bearish Sentiment
Shares of Mineralys Therapeutics (MLYS.O) declined sharply today, crossing below its 20-day simple moving average (SMA) as investor sentiment turned bearish. The stock's recent closing price of $47.18 reflects a projected downside, with Wells Fargo maintaining an Overweight rating yet setting a price target of $44.62, indicating a potential 5.42% decline.
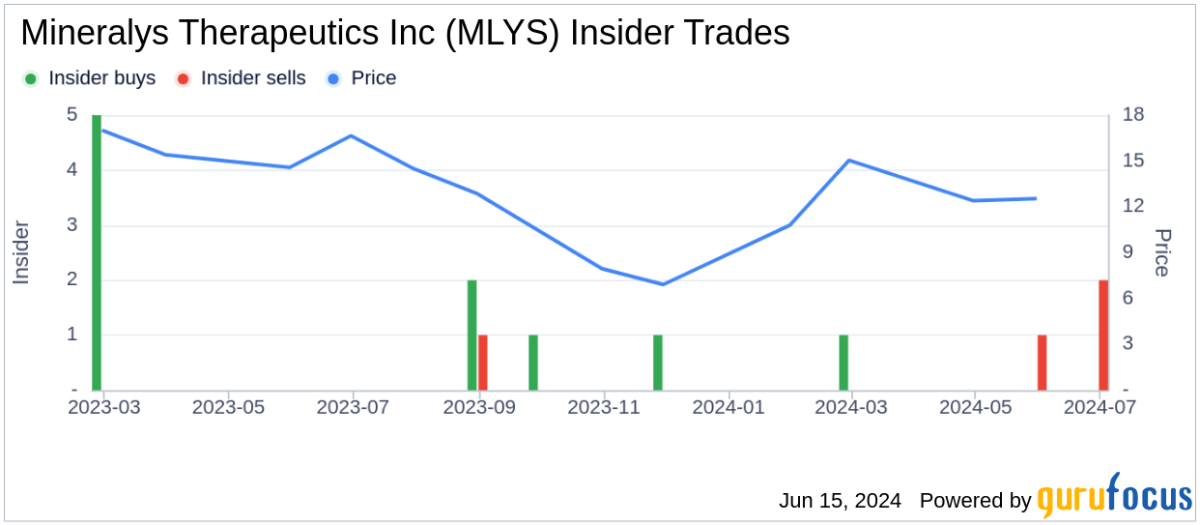
The technical breach of the 20-day SMA signals increased bearish momentum, further supported by a put/call ratio of 2.98, suggesting that investors are hedging against further declines. Despite a 5.14% increase in institutional ownership over the last quarter, which now totals 73,091K shares, major shareholders such as Catalys Pacific and Ra Capital Management have not adjusted their positions, indicating a cautious approach amidst market volatility.
As 327 funds report holdings in Mineralys, the lack of changes among top investors may reflect uncertainty in the stock's short-term performance, particularly in light of the current market outlook.
Trade with 70% Backtested Accuracy
Analyst Views on MLYS
About MLYS
About the author

- Stake Reduction: According to an SEC filing dated May 15, 2026, Velan Capital sold 140,000 shares of Mineralys Therapeutics in Q1, with an estimated transaction value of $4.11 million, indicating a cautious outlook on the company's future prospects.
- Decline in Position Value: Following the sale, Velan Capital's position value decreased by $5.18 million, reflecting the dual impact of trading activities and stock price changes, suggesting a weakening market confidence in Mineralys.
- Current Holdings: As of quarter-end, Velan Capital holds 11,000 shares of Mineralys valued at $297,990, with this stake now representing only 0.19% of its total assets under management, thus falling outside its top five holdings.
- Financial Status Analysis: Mineralys reported a loss of $39.3 million in Q1, but with $646 million in cash on hand, it should be able to sustain operations while the FDA reviews its lorundrostat application, although the lack of approved products poses ongoing risks.
- Share Reduction Details: According to an SEC filing dated May 15, 2026, Velan Capital sold 140,000 shares of Mineralys Therapeutics in Q1, valued at approximately $4.11 million, leaving it with 11,000 shares worth about $297,990, resulting in a net position decline of $5.18 million due to both share sales and price fluctuations.
- Holding Proportion Change: Following this reduction, Mineralys Therapeutics now represents only 0.19% of Velan Capital's reportable U.S. equity AUM, indicating a waning confidence in the company, while its top holdings include PRAX, TARA, and STRO.
- Market Performance Analysis: As of May 14, 2026, Mineralys Therapeutics shares were priced at $27.27, reflecting an 80.5% increase over the past year, significantly outperforming the S&P 500 by 53.18 percentage points, showcasing its strong position in the biopharmaceutical market.
- FDA Review Progress: Mineralys's lead product candidate, lorundrostat, is currently under FDA review, with an expected approval decision by December 22, 2026, which could present significant market opportunities for the company, despite reporting a loss of $39.3 million in Q1 and having no approved products yet.
- Clinical Trial Results: In a study involving 800 participants, lorundrostat achieved a significant reduction of 9.6 mmHg in systolic blood pressure among chronic kidney disease patients, highlighting its potential in managing hypertension and CKD effectively.
- Urinary Albumin Reduction: Among 84 participants with baseline albuminuria, lorundrostat treatment led to a remarkable 52.2% reduction in urinary albumin-to-creatinine ratio after 12 weeks, indicating its potential for renal protection and improving long-term patient outcomes.
- Safety Profile: Over the 12-week study, lorundrostat demonstrated a low incidence of confirmed hyperkalemia at 2.4% in CKD patients, showcasing its favorable safety profile and enhancing its attractiveness as a treatment option.
- FDA Review Progress: Lorundrostat is currently under review by the U.S. FDA, with results expected by December 22, 2026, which could open new market opportunities for the company to address the unmet needs of hypertension patients.
- FDA Acceptance Milestone: Mineralys Therapeutics successfully received FDA acceptance for the NDA of lorundrostat in Q1 2026, with a PDUFA target date set for December 22, 2026, establishing a crucial foundation for future commercialization efforts.
- Stable Financial Position: As of March 31, 2026, the company reported cash and cash equivalents totaling $646.1 million, indicating that these resources are expected to support operations into 2028, reflecting strong financial health.
- Decline in R&D Expenses: R&D expenses for Q1 2026 were $24.4 million, significantly down from $37.9 million in Q1 2025, showcasing a strategic shift in spending during the clinical trial phase.
- Market Access Strategy: Management emphasized a focus on market access for fourth-line hypertension patients, ensuring “parity access” with competitors to enhance the market competitiveness of lorundrostat.
- Stock Option Grants: On May 4, 2026, Mineralys Therapeutics granted a total of 29,760 stock options to two new non-executive employees, aimed at attracting and retaining talent, thereby enhancing the company's competitive edge in the biopharmaceutical sector.
- Restricted Stock Units: The company also awarded 22,320 restricted stock units that will vest over four years, with the first vesting occurring on the anniversary of the grant date, reflecting the company's long-term commitment to new employees and promoting team stability.
- Vesting Mechanism: The stock options will vest gradually over four years, with 25% vesting in the first year and 1/48 vesting monthly thereafter, ensuring that employees contribute continuously during their tenure, which enhances their sense of belonging.
- Compliance Assurance: This grant complies with Nasdaq Listing Rule 5635(c)(4), demonstrating the company's adherence to regulations and transparency in attracting new employees, which further boosts investor confidence in the company.
- Executive Stock Sale: Jon Congleton, CEO of Mineralys Therapeutics, sold 75,000 shares on March 31 through multiple open-market transactions valued at approximately $1.97 million, indicating active trading behavior in the company's stock.
- Holding Proportion Change: The sale impacted 9.61% of Congleton's direct holdings, leaving him with 705,051 shares, which represents about 58.6% of his position from August 2023, suggesting he still retains confidence in the company.
- Trading Plan Context: The stock sale was executed under Congleton's Rule 10b5-1 trading plan adopted in December 2025, which is designed to avoid legal issues related to insider trading, reflecting his cautious approach to compliance.
- Market Reaction and Risks: While Congleton's sale may raise market concerns, Mineralys stock has significantly dropped from its 52-week high of $47.65 last November in 2026, prompting investors to carefully assess the impact of the company's clinical trial results on future stock performance.