REGENXBIO Announces Positive Interim Data for RGX-202 in Duchenne Muscular Dystrophy Trial
Written by Emily J. Thompson, Senior Investment Analyst
Updated: 1 hour ago
0mins
Should l Buy RGNX?
Source: PRnewswire
- Clinical Trial Progress: REGENXBIO has announced positive interim data from the Phase I/II AFFINITY DUCHENNE trial of RGX-202, indicating significant improvements in functionality, safety, and biomarkers, positioning it as a potential best-in-class gene therapy for Duchenne muscular dystrophy.
- Expert Data Presentation: The trial's principal investigator, Dr. Carolina Tesi-Rocha, Clinical Professor of Neurology at Stanford University School of Medicine, presented these findings at the Muscular Dystrophy Association (MDA) Clinical and Scientific Conference, enhancing the therapy's academic recognition and industry visibility.
- Cardiac MRI Assessments: The new data includes cardiac MRI evaluations, suggesting RGX-202's potential in improving cardiac function, which is crucial for the overall health of Duchenne muscular dystrophy patients and may influence future treatment protocols.
- Optimistic Market Outlook: With positive clinical feedback, REGENXBIO is poised to carve out a niche in the competitive gene therapy market, attracting investor interest and potentially driving up the company's stock price.
Trade with 70% Backtested Accuracy
Stop guessing "Should I Buy RGNX?" and start using high-conviction signals backed by rigorous historical data.
Sign up today to access powerful investing tools and make smarter, data-driven decisions.
Analyst Views on RGNX
Wall Street analysts forecast RGNX stock price to rise
7 Analyst Rating
7 Buy
0 Hold
0 Sell
Strong Buy
Current: 9.900
Low
19.00
Averages
29.71
High
45.00
Current: 9.900
Low
19.00
Averages
29.71
High
45.00
About RGNX
REGENXBIO Inc. is a clinical-stage biotechnology company seeking to improve lives through the curative potential of gene therapy. The Company has developed a pipeline of gene therapy programs using its proprietary adeno-associated virus (AAV) gene therapy delivery platform (NAV Technology Platform) to address genetic diseases. It is focused on its internal development pipeline in three areas: retinal, neuromuscular, and neurodegenerative diseases. Its investigational AAV therapeutics include ABBV-RGX-314, RGX-202, RGX-121, and RGX-111. It is developing ABBV-RGX-314 in collaboration with AbbVie to treat large patient populations impacted by wet age-related macular degeneration, diabetic retinopathy (DR) and other chronic retinal diseases characterized by loss of vision. It is developing RGX-202 to treat Duchenne muscular dystrophy (Duchenne). The Company is developing RGX-121 to treat Mucopolysaccharidosis type II (MPS II), a progressive, neurodegenerative lysosomal storage disorder.
About the author

Emily J. Thompson
Emily J. Thompson, a Chartered Financial Analyst (CFA) with 12 years in investment research, graduated with honors from the Wharton School. Specializing in industrial and technology stocks, she provides in-depth analysis for Intellectia’s earnings and market brief reports.
- Clinical Trial Progress: REGENXBIO has announced positive interim data from the Phase I/II AFFINITY DUCHENNE trial of RGX-202, indicating significant improvements in functionality, safety, and biomarkers, positioning it as a potential best-in-class gene therapy for Duchenne muscular dystrophy.
- Expert Data Presentation: The trial's principal investigator, Dr. Carolina Tesi-Rocha, Clinical Professor of Neurology at Stanford University School of Medicine, presented these findings at the Muscular Dystrophy Association (MDA) Clinical and Scientific Conference, enhancing the therapy's academic recognition and industry visibility.
- Cardiac MRI Assessments: The new data includes cardiac MRI evaluations, suggesting RGX-202's potential in improving cardiac function, which is crucial for the overall health of Duchenne muscular dystrophy patients and may influence future treatment protocols.
- Optimistic Market Outlook: With positive clinical feedback, REGENXBIO is poised to carve out a niche in the competitive gene therapy market, attracting investor interest and potentially driving up the company's stock price.
See More

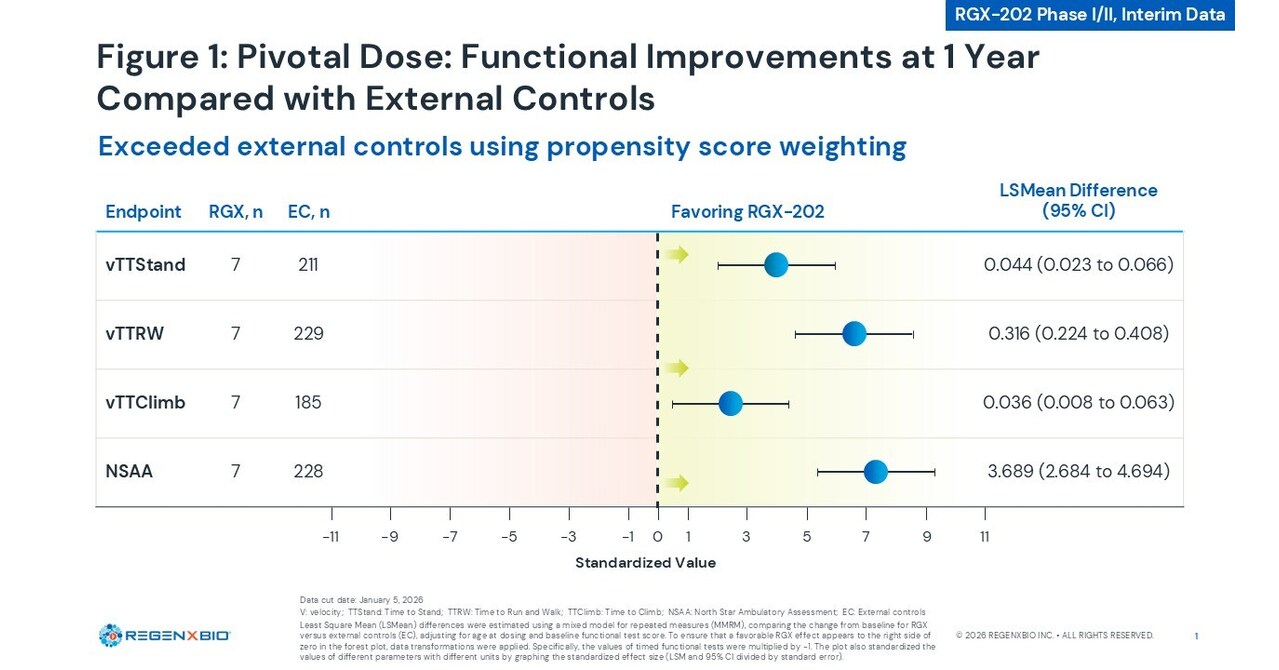
- Significant Functional Improvement: In the Phase I/II trial for Duchenne muscular dystrophy, RGX-202 demonstrated an average increase of 4.9 points on the North Star Ambulatory Assessment (NSAA) among seven participants at pivotal doses, particularly highlighting the efficacy in participants aged 8 and above, indicating the therapy's potential to positively alter disease progression.
- Stable Cardiac Health: Cardiac MRI data from pivotal dose participants showed stable left ventricular ejection fraction and global circumferential strain at one year post-treatment, underscoring RGX-202's safety and efficacy in maintaining cardiac function, which is strategically significant for its therapeutic profile.
- Favorable Safety Profile: The Phase I/II study reported no serious adverse events, with liver function markers remaining within normal limits for all participants, demonstrating RGX-202's good tolerability, likely attributed to its innovative microdystrophin construct and immune modulation strategy.
- Positive Future Outlook: REGENXBIO plans to release pivotal topline data in Q2 2026 and intends to hold a pre-BLA meeting with the FDA in mid-2026, reflecting the company's strong confidence in RGX-202's market potential as a meaningful treatment option for patients with Duchenne muscular dystrophy.
See More
Company Overview: Regenxbio is a biotechnology company focused on gene therapy, particularly in the treatment of various diseases.
Upcoming Data Release: The company is expected to release pivotal top-line data in the second quarter of 2026, which could significantly impact its future prospects.
See More
- Legal Investigation Launched: Faruq & Faruqi LLP is investigating potential claims against REGENXBIO, focusing on investors who purchased or acquired securities between February 9, 2022, and January 27, 2026, highlighting the firm's commitment to investor rights.
- Investor Contact Information: The firm encourages affected investors to reach out directly to partner Josh Wilson, providing contact numbers 877-247-4292 and 212-983-9330 (Ext. 1310), ensuring timely legal support for those impacted.
- Class Action Reminder: Faruq & Faruqi reminds investors that April 14, 2026, is the deadline to seek lead plaintiff status in a federal securities class action against REGENXBIO, emphasizing the urgency of the legal process.
- Role of Securities Law Firm: As a leading national securities law firm, Faruq & Faruqi's investigation indicates potential legal liabilities for REGENXBIO, urging investors to be vigilant about protecting their investment rights.
See More
- Class Action Notification: The Schall Law Firm reminds investors of a class action lawsuit against Regenxbio for violations of securities laws, involving securities transactions from February 9, 2022, to January 27, 2026, with a deadline for participation set for April 14, 2026.
- False Statements Exposed: The complaint alleges that Regenxbio made overly optimistic claims about its product candidate RGX-111 while concealing negative data regarding its efficacy and safety, resulting in significant investor losses once the truth was revealed.
- Tumor Discovery Incident: An intraventricular CNS tumor was discovered in a participant during the RGX-111 study, and the disclosure of this information rendered Regenxbio's public statements false and materially misleading, exacerbating investor losses.
- Legal Representation Services: The Schall Law Firm specializes in securities class action lawsuits and offers free consultations to affected investors, emphasizing its commitment to protecting shareholder rights and assisting in loss recovery.
See More
- Class Action Filed: Bronstein, Gewirtz & Grossman, LLC has initiated a class action lawsuit against REGENXBIO, alleging violations of federal securities laws from February 9, 2022, to January 27, 2026, indicating significant investor concern over the company's transparency and accountability.
- Allegations of Misrepresentation: The complaint claims that REGENXBIO made materially misleading statements regarding the development of RGX-111 gene therapy, particularly that optimistic projections about its clinical trial lacked a reasonable basis, potentially misleading investors about the company's future prospects.
- Investor Rights Protection: Affected investors are encouraged to apply to be lead plaintiffs by April 14, 2026, highlighting the legal team's commitment to safeguarding investor rights while emphasizing the potential impact of the class action on REGENXBIO's accountability.
- Contingency Fee Arrangement: Bronstein, Gewirtz & Grossman, LLC will represent investors on a contingency fee basis, meaning they will only charge fees if they successfully recover damages, thereby reducing financial risk for investors and enhancing the appeal of participating in the lawsuit.
See More









