ImmunityBio Faces Securities Fraud Lawsuit
- Lawsuit Background: Block & Leviton has announced a securities fraud lawsuit against ImmunityBio and certain executives, alleging that false or misleading statements made between January 19, 2026, and March 24, 2026, led to investor losses.
- False Statements: The lawsuit claims that Patrick Soon-Shiong materially overstated Anktiva's capabilities, resulting in materially false and misleading statements regarding ImmunityBio's business and operations, causing investor damages when the truth emerged.
- Investor Eligibility: Investors who purchased ImmunityBio common stock during the class period and suffered losses are encouraged to contact Block & Leviton to learn how to seek recovery, with a deadline of May 26, 2026, for lead plaintiff applications.
- Whistleblower Program: Individuals with non-public information about ImmunityBio are encouraged to assist in the investigation or file a report with the SEC, with potential rewards of up to 30% of any successful recovery for whistleblowers.
Trade with 70% Backtested Accuracy
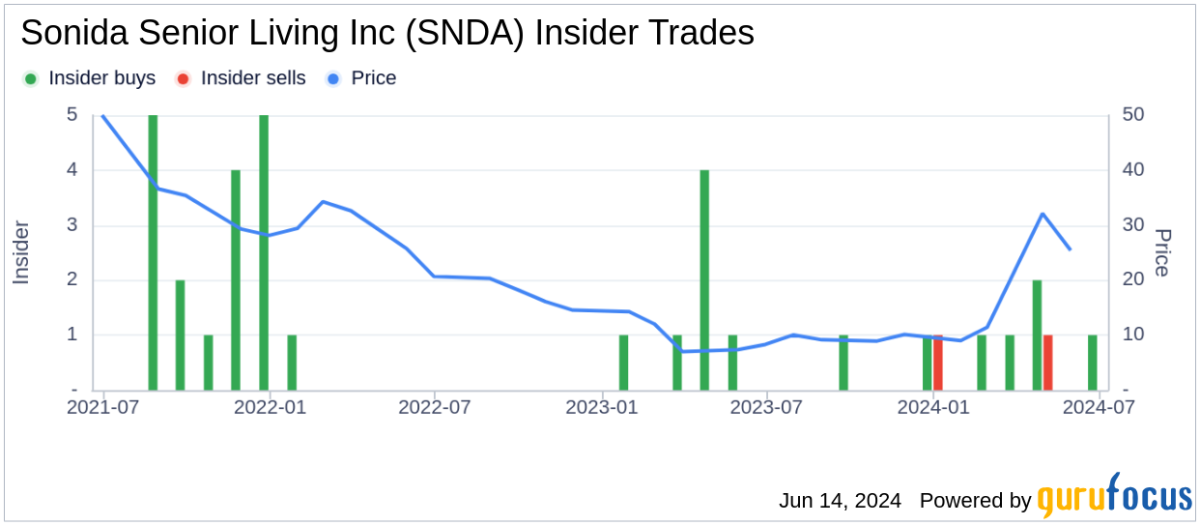
Analyst Views on IBRX
About IBRX
About the author

Community Bio Initiative: The initiative focuses on submitting supplemental information to enhance community engagement and support.
Contractual Framework: The project is structured under a contract that outlines the responsibilities and expectations for all parties involved.
Timeline for Submission: The goal is to have all supplemental materials submitted by the year 2026.
Impact on Local Development: The initiative aims to foster local development and improve community resources through collaborative efforts.

Bladder Cancer Study Update: A Phase 3 bladder cancer study has reached a key efficacy endpoint, prompting a review by an independent monitoring committee. The trial is comparing a combination therapy of Anktiva and BCG against BCG alone in newly diagnosed patients.
Stock Performance: Shares of Immunity Bio (IBRX) surged by about 9% following the announcement of new trials and progress in their bladder cancer study, recovering from a previous sharp sell-off.
New Trials for Severe Pneumonia: Immunity Bio has initiated new studies evaluating Anktiva in combination with off-the-shelf immune cell therapy for critically ill patients with severe pneumonia, aiming to improve immune function and survival rates.
Market Sentiment: Retail sentiment for IBRX has shifted positively, with significant increases in message volumes, indicating growing investor interest and optimism about the company's potential in cancer therapies.
- FDA Action: The Food and Drug Administration (FDA) accused ImmunityBio of using false or misleading advertising for its bladder cancer drug, Anktiva.
- Market Reaction: Following the FDA's announcement, shares of ImmunityBio experienced a significant decline.
- Lawsuit Background: Block & Leviton has announced a securities fraud lawsuit against ImmunityBio and certain executives, alleging that false or misleading statements made between January 19, 2026, and March 24, 2026, led to investor losses.
- False Statements: The lawsuit claims that Patrick Soon-Shiong materially overstated Anktiva's capabilities, resulting in materially false and misleading statements regarding ImmunityBio's business and operations, causing investor damages when the truth emerged.
- Investor Eligibility: Investors who purchased ImmunityBio common stock during the class period and suffered losses are encouraged to contact Block & Leviton to learn how to seek recovery, with a deadline of May 26, 2026, for lead plaintiff applications.
- Whistleblower Program: Individuals with non-public information about ImmunityBio are encouraged to assist in the investigation or file a report with the SEC, with potential rewards of up to 30% of any successful recovery for whistleblowers.
- Market Growth Potential: The global cell therapy manufacturing market is projected to exceed $7 billion in 2026 and reach $14 billion by 2035, indicating strong growth momentum that drives strategic positioning for companies like Avaí Bio.
- Regulatory Flexibility Enhancement: The FDA's easing of manufacturing requirements for cell and gene therapies accelerates the approval timeline for life-saving products, creating a historic shift that provides companies with scalable bioproduction infrastructure and enhances market competitiveness.
- Cell Bank Development: Avaí Bio, in partnership with Austrianova, has begun manufacturing a Master Cell Bank (MCB) for α-Klotho protein, establishing a stable production foundation that ensures product quality and consistency for future cell therapies.
- Clinical Trial Outlook: The production of α-Klotho cells will directly support Austrianova's Cell-in-a-Box® platform, aiming to provide patients with durable anti-aging treatments, marking a significant advancement for the company in addressing aging-related diseases.
- Market Growth: The global cell therapy manufacturing market is projected to exceed $7 billion in 2026 and reach $14 billion by 2035, indicating robust growth potential that drives demand for cell and gene therapies.
- FDA Regulatory Impact: The FDA's easing of manufacturing requirements accelerates the approval timeline for life-saving products, creating sustainable value for therapy developers like Avaí Bio, marking a historic shift in the industry.
- Strategic Production Capacity: As biologics production capacity becomes a coveted strategic asset, Avaí Bio and Austrianova have commenced manufacturing a Master Cell Bank of α-Klotho protein, ensuring high quality and consistency for future production.
- Innovative Technology Application: Avaí Bio's cell therapy products will be developed using the Cell-in-a-Box® encapsulation platform, aiming to provide durable anti-aging treatments for patients, signifying the company's successful pivot into biotechnology.