Annovis Biopress Reports FY GAAP EPS of -$1.40
- Earnings Report: Annovis Biopress reported a FY GAAP EPS of -$1.40, indicating challenges in profitability that may affect investor confidence and market perception.
- Cash Position: As of December 31, 2025, Annovis had cash and cash equivalents totaling $19.5 million, a significant increase from $10.6 million in 2024, suggesting improved financial management.
- Funding Outlook: The company estimates that its current cash and equivalents, including proceeds from stock offerings in Q4 2025, will fund operations into Q3 2026, indicating a degree of financial stability.
- Share Count: Annovis had 27.2 million shares of common stock outstanding as of December 31, 2025, reflecting the company's activity in the capital markets and the stability of its shareholder structure.
Trade with 70% Backtested Accuracy
Analyst Views on ANVS
About ANVS
About the author

- Patent Protection Expansion: Annovis Bio announced the issuance of U.S. Patent No. 12,582,632 B2, covering the prevention and treatment of neurological injuries from brain infections through the use of buntanetap, showcasing the company's innovative potential in neurodegenerative diseases.
- Scientific Research Support: Studies indicate that infections from viruses and bacteria may be at the root of Alzheimer's disease, with buntanetap targeting the biological process by reducing the overproduction of neurotoxic proteins, potentially offering new treatment options for patients.
- Broad Market Prospects: The patent encompasses preventive use in healthy individuals and reversal of neurological damage in those already affected, reflecting the company's strategic positioning against various microbial threats, which is expected to enhance its competitive edge in the market.
- Long-Term Protection Advantage: The new patent protection extends through 2044, ensuring Annovis's market exclusivity for several years, further solidifying its leadership position in the biotechnology sector.
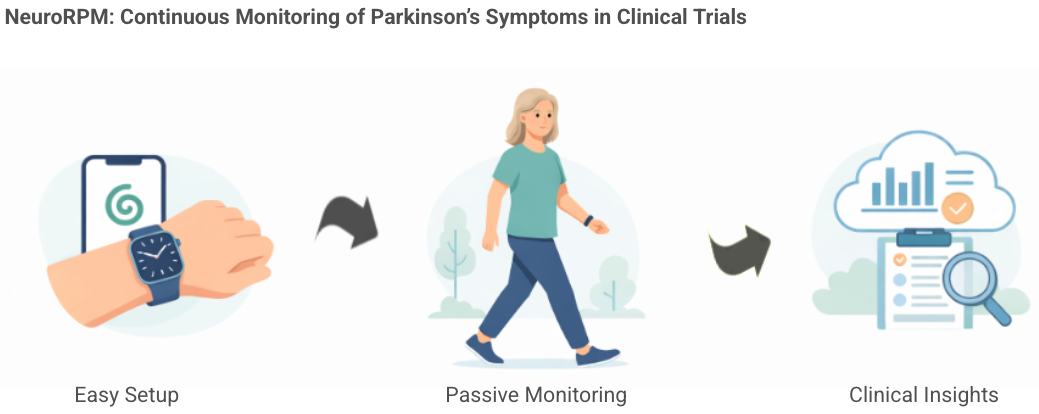
- Clinical Trial Collaboration: NeuroRPM's FDA-cleared wearable platform will be utilized in Annovis Bio's ANVS-25002 Parkinson's disease clinical trial, expected to enroll around 500 patients, aiming to enhance treatment outcomes through real-time monitoring.
- Data Collection Innovation: The platform leverages the Apple Watch to passively collect movement data, providing continuous measurement of key Parkinson's motor symptoms, thereby assisting researchers in more accurately assessing treatment responses.
- AI Technology Application: NeuroRPM's AI-enabled monitoring device analyzes complex symptom patterns, enhancing understanding of symptom fluctuations in patients' daily lives, thus providing a richer evidence base for clinical research.
- Long-term Monitoring Commitment: Following the trial, participants may continue using NeuroRPM for ongoing clinical monitoring, indicating the platform's long-term application potential in Parkinson's disease management, helping patients achieve better treatment experiences.
- Earnings Report: Annovis Biopress reported a FY GAAP EPS of -$1.40, indicating challenges in profitability that may affect investor confidence and market perception.
- Cash Position: As of December 31, 2025, Annovis had cash and cash equivalents totaling $19.5 million, a significant increase from $10.6 million in 2024, suggesting improved financial management.
- Funding Outlook: The company estimates that its current cash and equivalents, including proceeds from stock offerings in Q4 2025, will fund operations into Q3 2026, indicating a degree of financial stability.
- Share Count: Annovis had 27.2 million shares of common stock outstanding as of December 31, 2025, reflecting the company's activity in the capital markets and the stability of its shareholder structure.
- Investor Attention: As the earnings season unfolds, mid to low market capitalization healthcare stocks are drawing investor attention due to their strong earnings momentum, indicating growing market confidence in this sector.
- Analyst Expectations: The EPS Revision Grade reflects the trend in analyst earnings estimates, with A+ ratings indicating optimistic projections for future performance, potentially driving stock prices higher.
- List of A+ Rated Stocks: Currently, companies such as Aldeyra Therapeutics, Altimmune, Annovis Bio, and Assertio Holdings have received A+ EPS Revision Grades, showcasing their strong performance in the eyes of analysts.
- Market Strategy Impact: These A+ rated healthcare stocks are likely to attract more investor interest, potentially triggering positive sentiment towards the healthcare sector as a whole, thereby enhancing the performance of related ETFs.

Long-term Study Launch: Annovis Bio, Inc. is set to begin a long-term open-label extension study in January 2026 to evaluate the effects of Buntanetap on Parkinson's disease patients over three years, aiming to enroll 500 participants across multiple U.S. sites.
Cohorts and Objectives: The study will include two cohorts: former participants from earlier studies and patients receiving deep brain stimulation, focusing on the long-term safety, efficacy, and potential disease-modifying properties of Buntanetap.
Regulatory Preparation: This extension study is part of Annovis' strategy to support a future New Drug Application, as it aims to meet FDA requirements for total patient numbers and treatment duration, building on previous studies involving over 1,200 patients.
Market Performance: Annovis' stock has fluctuated between $1.11 and $5.60 over the past year, closing at $3.67 and rising to $3.79 in overnight trading, reflecting investor interest in the company's developments in Parkinson's disease treatment.
- Study Launch: Annovis Bio announced the initiation of a 36-month Open-Label Extension study starting January 2026, aiming to enroll 500 Parkinson's patients to evaluate the long-term safety and efficacy of the drug buntanetap, thereby providing continued treatment access for patients.
- Patient Populations: The study will include former clinical trial participants and patients receiving deep brain stimulation, aiming to assess the effects of buntanetap in different treatment contexts, addressing a significant gap in clinical research for underserved populations.
- Biomarker Collection: Throughout the study, skin and plasma biomarkers will be collected to deepen the understanding of disease progression and treatment effects, aiding the company in comprehending buntanetap's potential as a disease-modifying therapy.
- NDA Readiness: This study will assist Annovis in meeting FDA patient exposure requirements, with total patient participation expected to reach approximately 1,500, ensuring the company is well-prepared for future New Drug Application submissions.