MiniMed Flex Receives FDA Clearance for Next-Gen Insulin Pump
Written by Emily J. Thompson, Senior Investment Analyst
Updated: 1 hour ago
0mins
Should l Buy MMED?
Source: seekingalpha
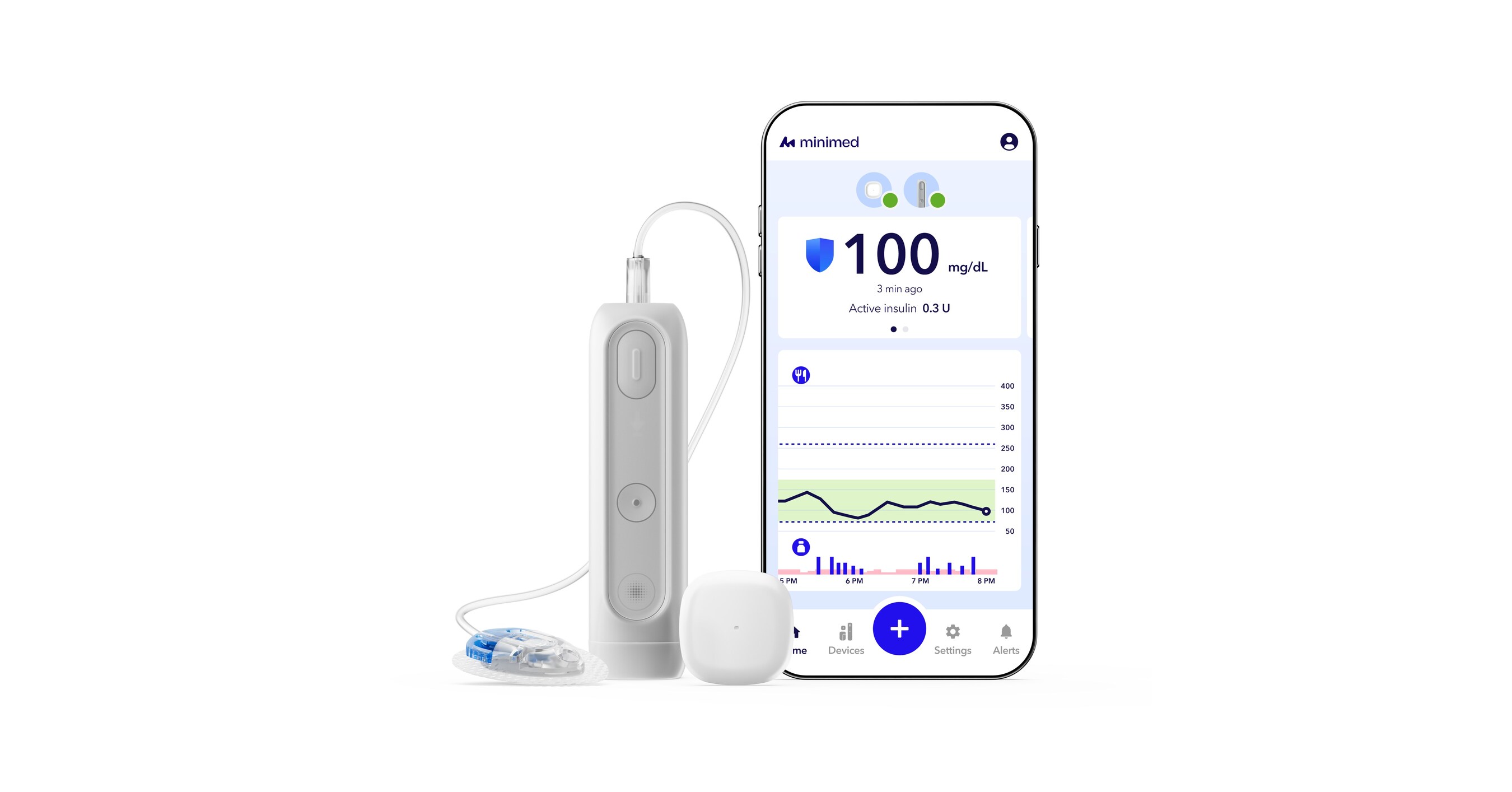
- FDA Approval: MiniMed announced that its next-generation smartphone-controlled insulin pump, MiniMed Flex, has received clearance from the U.S. Food and Drug Administration (FDA), marking a significant advancement in diabetes management devices.
- Product Design: The MiniMed Flex is about half the size of the MiniMed 780G, featuring a discreet design aimed at providing a more intuitive and lifestyle-friendly management solution for diabetes patients, highlighting the company's focus on user experience.
- Market Launch Plan: MiniMed plans to initiate a customer experience phase this spring with select current customers, followed by a broader commercial launch in the summer, which is expected to enhance market penetration.
- Stock Price Reaction: MiniMed's shares rose 1% in premarket trading, reflecting a positive market response to the new product launch, which may bolster investor confidence in the company's future growth potential.
Trade with 70% Backtested Accuracy
Stop guessing "Should I Buy MMED?" and start using high-conviction signals backed by rigorous historical data.
Sign up today to access powerful investing tools and make smarter, data-driven decisions.
Analyst Views on MMED
Wall Street analysts forecast MMED stock price to rise
0 Analyst Rating
0 Buy
0 Hold
0 Sell
Current: 16.830
Low
Averages
High
Current: 16.830
Low
Averages
High

No data
About MMED
MiniMed Group, Inc. is a global medical technology company that develops, manufactures, and markets a suite of solutions for the management of diabetes. The Company's fully integrated system addresses two key pain points for PWD: health outcomes and complexity of diabetes management. The MiniMed 780G is its flagship AID system, which consists of the components: the MiniMed 780G insulin pump, its second-generation insulin pump offering enhanced functionality and technological integration with other components of its 780G system; the Simplera Sync or Instinct CGM sensor, which are both easy-to-use, fully disposable, two-step insertion sensors; and its SmartGuard dosing algorithm technology, which automatically delivers basal insulin and auto-correction doses every five minutes based on sensor glucose readings and adapts to ongoing changes in user behavior patterns every night. For PWD that choose to manage their diabetes with MDI instead of an insulin pump, it offers Smart MDI system.
About the author

Emily J. Thompson
Emily J. Thompson, a Chartered Financial Analyst (CFA) with 12 years in investment research, graduated with honors from the Wharton School. Specializing in industrial and technology stocks, she provides in-depth analysis for Intellectia’s earnings and market brief reports.
- Product Innovation: MiniMed Flex™, approximately half the size of the MiniMed™ 780G system, features a screenless design and the SmartGuard™ adaptive algorithm that adjusts insulin delivery in real-time, aiming to enhance the daily management experience for diabetes patients.
- FDA Approval Milestone: Securing FDA clearance just one week after the company's IPO marks a significant milestone for MiniMed as an independent public company, demonstrating its commitment to innovation and is expected to drive market share growth.
- User Upgrade Program: The launch of the MiniMed™ Forward Program allows existing MiniMed™ 780G users to upgrade to MiniMed Flex™ at no cost, a strategy that not only enhances customer loyalty but also promotes market acceptance of the new product.
- Improved Clinical Performance: Equipped with Meal Detection™ technology, MiniMed Flex™ has proven clinical performance that exceeds global Time in Range recommendations, which is expected to improve clinical outcomes for diabetes patients and enhance their quality of life.
See More
- FDA Approval: MiniMed announced that its next-generation smartphone-controlled insulin pump, MiniMed Flex, has received clearance from the U.S. Food and Drug Administration (FDA), marking a significant advancement in diabetes management devices.
- Product Design: The MiniMed Flex is about half the size of the MiniMed 780G, featuring a discreet design aimed at providing a more intuitive and lifestyle-friendly management solution for diabetes patients, highlighting the company's focus on user experience.
- Market Launch Plan: MiniMed plans to initiate a customer experience phase this spring with select current customers, followed by a broader commercial launch in the summer, which is expected to enhance market penetration.
- Stock Price Reaction: MiniMed's shares rose 1% in premarket trading, reflecting a positive market response to the new product launch, which may bolster investor confidence in the company's future growth potential.
See More
- Product Innovation: MiniMed Flex™, the next-generation insulin pump, is half the size of the MiniMed™ 780G system and features a screenless design, making it easier for users to integrate into their daily lives, representing a significant advancement in insulin pump technology.
- Smart Algorithm Implementation: The system is powered by the SmartGuard™ adaptive algorithm with Meal Detection™ technology, which has clinical performance exceeding global Time in Range recommendations, expected to significantly enhance daily management efficiency and quality of life for diabetes patients.
- User Upgrade Program: The MiniMed™ Forward Program allows existing MiniMed™ 780G users to upgrade to MiniMed Flex™ at no cost, which not only increases user retention but also demonstrates the company's commitment to customer experience.
- Market Launch Strategy: MiniMed Flex™ will undergo a customer experience phase in spring, followed by a broader commercial launch in summer, anticipated to attract more diabetes patients and further solidify MiniMed's leadership position in the market.
See More
- Successful IPO: MiniMed Group, Inc. completed its IPO on March 6, 2026, issuing 28 million shares at $20 each, raising approximately $538 million, which significantly strengthens the company's capital base for future growth initiatives.
- Equity Structure Change: Following the IPO, Medtronic plc retains about 90.03% ownership of MiniMed, indicating Medtronic's strong control over MiniMed and potentially paving the way for a future split-off, which could impact strategic direction.
- Strong Underwriter Lineup: The IPO was led by Goldman Sachs, BofA Securities, Citigroup, and Morgan Stanley, with several other reputable financial institutions involved, reflecting market confidence in MiniMed and its potential in the healthcare sector.
- Clear Use of Proceeds: MiniMed intends to allocate part of the proceeds for general corporate purposes while using the remainder to repay intercompany debt owed to Medtronic, a move that will help optimize its financial structure and enhance financial flexibility.
See More
- MiniMed IPO Performance: MiniMed Group (MMED) priced its IPO below expectations, raising $560 million at a $5.6 billion market cap, and despite holding a significant share in insulin delivery and continuous glucose monitoring markets, it faced competitive pressures, finishing down 8% on its first day.
- Overall Market Performance: As of March 5, 2026, the Renaissance IPO Index is down 5.2% year-to-date, indicating weakness in the IPO market, while the S&P 500 remains flat, reflecting investor caution towards new public offerings.
- SPAC Activity Overview: This week, alongside MiniMed, two SPACs, Kensington Capital VI and GalaxyEdge Acquisition, went public, both failing to generate investor enthusiasm with first-day returns of 0%, highlighting a lack of interest in blank check companies.
- Robinhood Fund Performance: Although not included in the IPO count, Robinhood Ventures Fund I (RVI) raised $315 million in its listing but saw a 16% drop in its share price, indicating insufficient market confidence in new funds.
See More
- IPO Financing Details: MiniMed Group raised $560 million through its IPO, yet opened at $19.05, reflecting a roughly 5% decline from the $20 offering price, indicating market caution regarding its prospects.
- Stock Performance Fluctuations: On its debut, MiniMed's stock further dropped to $18.95 shortly after opening, reflecting investor concerns about its market positioning and future profitability, which could impact the company's future financing capabilities.
- Market Valuation: Post-IPO, MiniMed is valued at $5.6 billion based on over 280.8 million shares outstanding, although this valuation fell short of market expectations, potentially limiting its future expansion plans.
- CEO Statement: MiniMed CEO Que Dallara remarked that today marks a defining moment for MiniMed as a standalone public company during the Nasdaq opening, and despite the lukewarm market response, the company must focus on its future strategic development.
See More