Kodiak Sciences' Zenkuda Therapy Achieves Key Milestones in Phase 3 Trial
Written by Emily J. Thompson, Senior Investment Analyst
Updated: 1 hour ago
0mins
Should l Buy KOD?
Source: seekingalpha
- Clinical Trial Success: Kodiak Sciences announced significant achievements in its Phase 3 trial for the eye disorder therapy Zenkuda, with nearly 63% of diabetic retinopathy patients showing a ≥2-step improvement in the diabetic retinopathy severity scale (DRSS) at 48 weeks, indicating strong statistical significance.
- Control Group Comparison: In contrast, only about 3% of subjects in the sham arm experienced a ≥2-step improvement in DRSS, highlighting Zenkuda's clear therapeutic advantage and its potential to reshape treatment paradigms for diabetic retinopathy.
- Good Safety Profile: The intravitreal injection of Zenkuda was well tolerated among patients, with a low incidence of common ocular adverse events, enhancing its appeal as a treatment option and potentially increasing market acceptance.
- Accelerated Application Plans: Following the positive GLOW2 results, Kodiak Sciences stated that it has a Biologics License Application (BLA)-ready profile and intends to expedite its submission plans, signaling potential commercial success in the near future.
Trade with 70% Backtested Accuracy
Stop guessing "Should I Buy KOD?" and start using high-conviction signals backed by rigorous historical data.
Sign up today to access powerful investing tools and make smarter, data-driven decisions.
Analyst Views on KOD
Wall Street analysts forecast KOD stock price to rise
7 Analyst Rating
5 Buy
2 Hold
0 Sell
Moderate Buy
Current: 21.850
Low
14.00
Averages
27.86
High
50.00
Current: 21.850
Low
14.00
Averages
27.86
High
50.00
About KOD
Kodiak Sciences Inc. is a biopharmaceutical company. The Company has developed a new technology platform, the Antibody Biopolymer Conjugate (ABC) Platform, for retinal medicines. Its ABC Platform uses molecular engineering to merge the fields of protein-based and chemistry-based therapies. Its lead investigational medicine, tarcocimab tedromer, is a novel anti-vascular endothelial growth factor (anti-VEGF) antibody biopolymer conjugate under development for the treatment of high prevalence retinal vascular diseases, including diabetic retinopathy and wet age-related macular degeneration. Its second investigational medicine, KSI-501, is a bispecific antibody biopolymer conjugate designed to inhibit interleukin-6 (IL-6)-mediated inflammation and VEGF. It is also developing a third investigational medicine, KSI-101, an unconjugated bispecific protein targeting IL-6 and VEGF.
About the author

Emily J. Thompson
Emily J. Thompson, a Chartered Financial Analyst (CFA) with 12 years in investment research, graduated with honors from the Wharton School. Specializing in industrial and technology stocks, she provides in-depth analysis for Intellectia’s earnings and market brief reports.

KODIAK Sciences Stock Surge: KODIAK Sciences shares experienced a significant increase of 40% in pre-market trading.
Success of Eye Drug Study: The surge in stock price follows positive results from a late-stage study of their eye drug, indicating its effectiveness.
See More
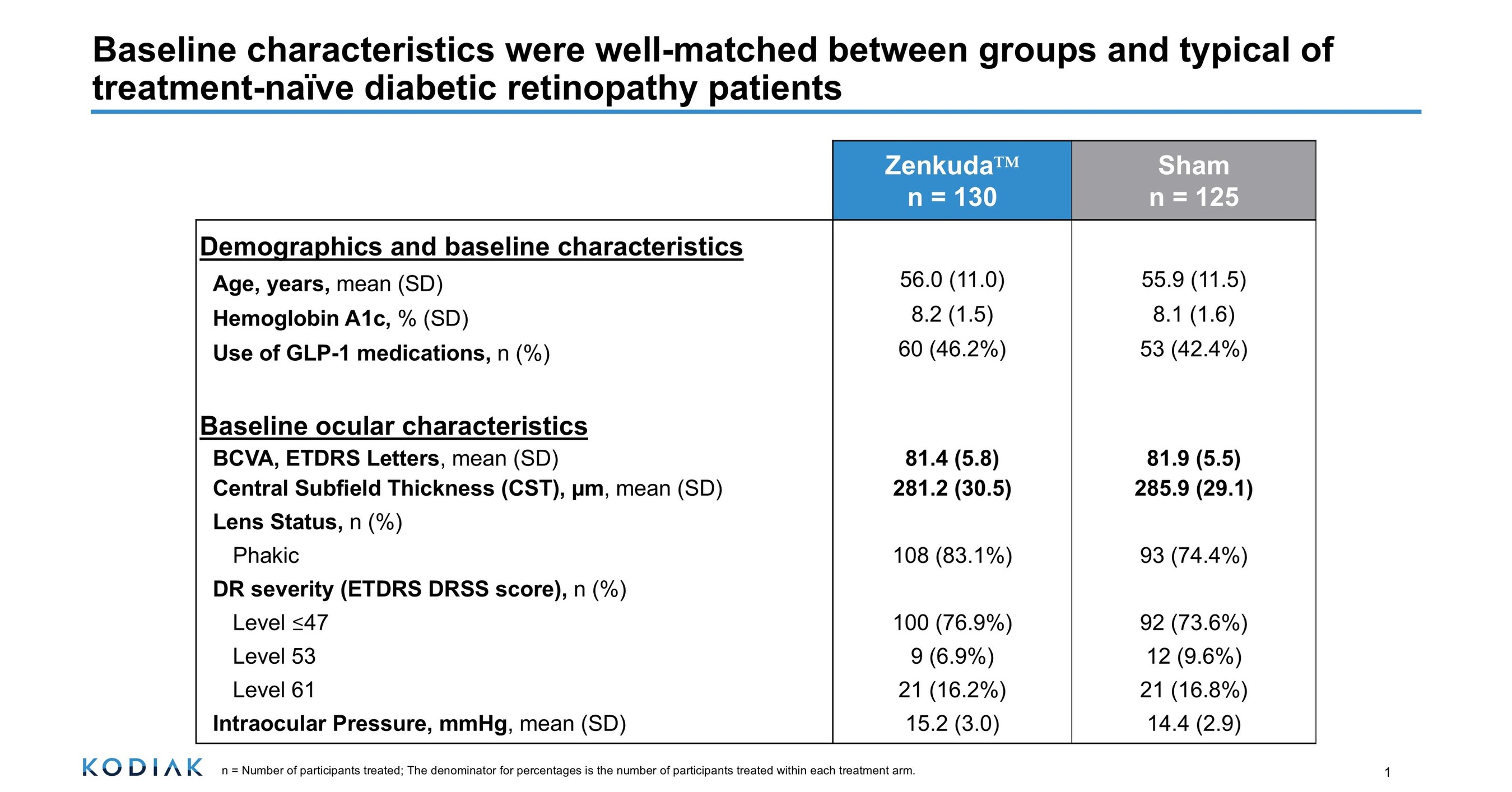
- Clinical Trial Success: Kodiak Sciences' GLOW2 Phase 3 study shows that 62.5% of diabetic retinopathy patients treated with Zenkuda achieved a ≥2-step improvement in DRSS score, significantly outperforming the 3.3% in the sham group, demonstrating strong efficacy and high statistical significance (p<0.0001).
- Good Safety Profile: Zenkuda exhibited excellent tolerability with low rates of common ocular adverse events, and notably, no cases of intraocular inflammation were reported, indicating a high safety profile that may facilitate broader clinical adoption.
- Flexible Dosing Intervals: The GLOW2 study employed a 6-month dosing interval, supporting flexible adjustments based on patient needs, which could transform treatment options for physicians and patients, enhancing adherence and therapeutic outcomes.
- Multi-Indication BLA Readiness: Kodiak Sciences intends to accelerate the BLA submission for Zenkuda, leveraging robust data from both GLOW1 and GLOW2 to showcase its potential in diabetic retinopathy and other indications, further expanding market opportunities.
See More

Key Findings: KODIAK SCIENCES demonstrated a significant 85% risk reduction in key secondary endpoints.
Comparison: The results indicate a superiority of KODIAK SCIENCES' treatment over SHAM.
See More
- Clinical Trial Success: In the GLOW2 study, 62.5% of patients treated with Zenkuda achieved a ≥2-step improvement in Diabetic Retinopathy Severity Score (DRSS), compared to only 3.3% in the control group, demonstrating significant clinical superiority (p<0.0001), which is likely to enhance Zenkuda's market acceptance and utilization.
- Key Secondary Endpoint Improvement: Zenkuda showed an impressive 85% reduction in the risk of developing sight-threatening complications, with an incidence of 2.4% compared to 15.8% in the control group (p=0.0001), highlighting its critical role in preventing disease progression.
- Favorable Safety Profile: The study reported a 0% rate of intraocular inflammation for Zenkuda, with a cataract adverse event rate of 2.3%, comparable to 1.6% in the control group, further reinforcing its safety and tolerability, which boosts confidence in its market promotion.
- Accelerated BLA Submission: Following the robust data from the GLOW2 study, Kodiak intends to expedite the Biologics License Application (BLA) for Zenkuda, paving the way for commercialization in the diabetic retinopathy space, which is expected to significantly enhance the company's future revenue potential.
See More
- Clinical Trial Success: Kodiak Sciences announced significant achievements in its Phase 3 trial for the eye disorder therapy Zenkuda, with nearly 63% of diabetic retinopathy patients showing a ≥2-step improvement in the diabetic retinopathy severity scale (DRSS) at 48 weeks, indicating strong statistical significance.
- Control Group Comparison: In contrast, only about 3% of subjects in the sham arm experienced a ≥2-step improvement in DRSS, highlighting Zenkuda's clear therapeutic advantage and its potential to reshape treatment paradigms for diabetic retinopathy.
- Good Safety Profile: The intravitreal injection of Zenkuda was well tolerated among patients, with a low incidence of common ocular adverse events, enhancing its appeal as a treatment option and potentially increasing market acceptance.
- Accelerated Application Plans: Following the positive GLOW2 results, Kodiak Sciences stated that it has a Biologics License Application (BLA)-ready profile and intends to expedite its submission plans, signaling potential commercial success in the near future.
See More
- Successful Clinical Trial: Kodiak Sciences reported positive Phase 3 results for its eye drug Zenkuda, with 62.5% of patients achieving at least a two-step improvement in DRSS score by week 48, compared to just 3.3% in the control group, indicating significant efficacy in treating diabetic retinopathy.
- Significant Vision Protection: Zenkuda reduced the risk of sight-threatening complications by 85% through Week 48, which not only enhances patient quality of life but also positions the company to capture a larger market share in the ophthalmic drug sector.
- Strong Performance Among GLP-1 Users: The drug demonstrated consistent benefits regardless of GLP-1 use, with 60% of patients on GLP-1 therapies achieving a two-step DRSS improvement, highlighting its broad applicability and potential market reach.
- Accelerated Submission Plans: CEO Victor Perlroth stated that Zenkuda now has a multi-indication BLA-ready profile and intends to expedite BLA submission plans, laying the groundwork for future growth and market expansion.
See More






