Precigen's PAPZIMEOS FDA Approval Drives Stock Surge
Precigen Inc's stock surged by 27.74% as it crossed above the 5-day SMA, reflecting strong market interest following the FDA approval of its product PAPZIMEOS.
The company reported that PAPZIMEOS received FDA approval for treating recurrent respiratory papillomatosis, marking a significant transition to commercial sales with first revenues of $3.4 million. This approval has garnered robust support from the medical community, with over 300 patient enrollments, indicating a successful market entry and potential for future growth. Additionally, the CEO projected optimistic revenue forecasts for Q1 2026, further boosting investor confidence.
This positive momentum suggests that Precigen is on a path to capitalize on its recent FDA approval, with expectations of increased market share and revenue growth, despite facing challenges in managing operational expenses.
Trade with 70% Backtested Accuracy
Analyst Views on PGEN
About PGEN
About the author

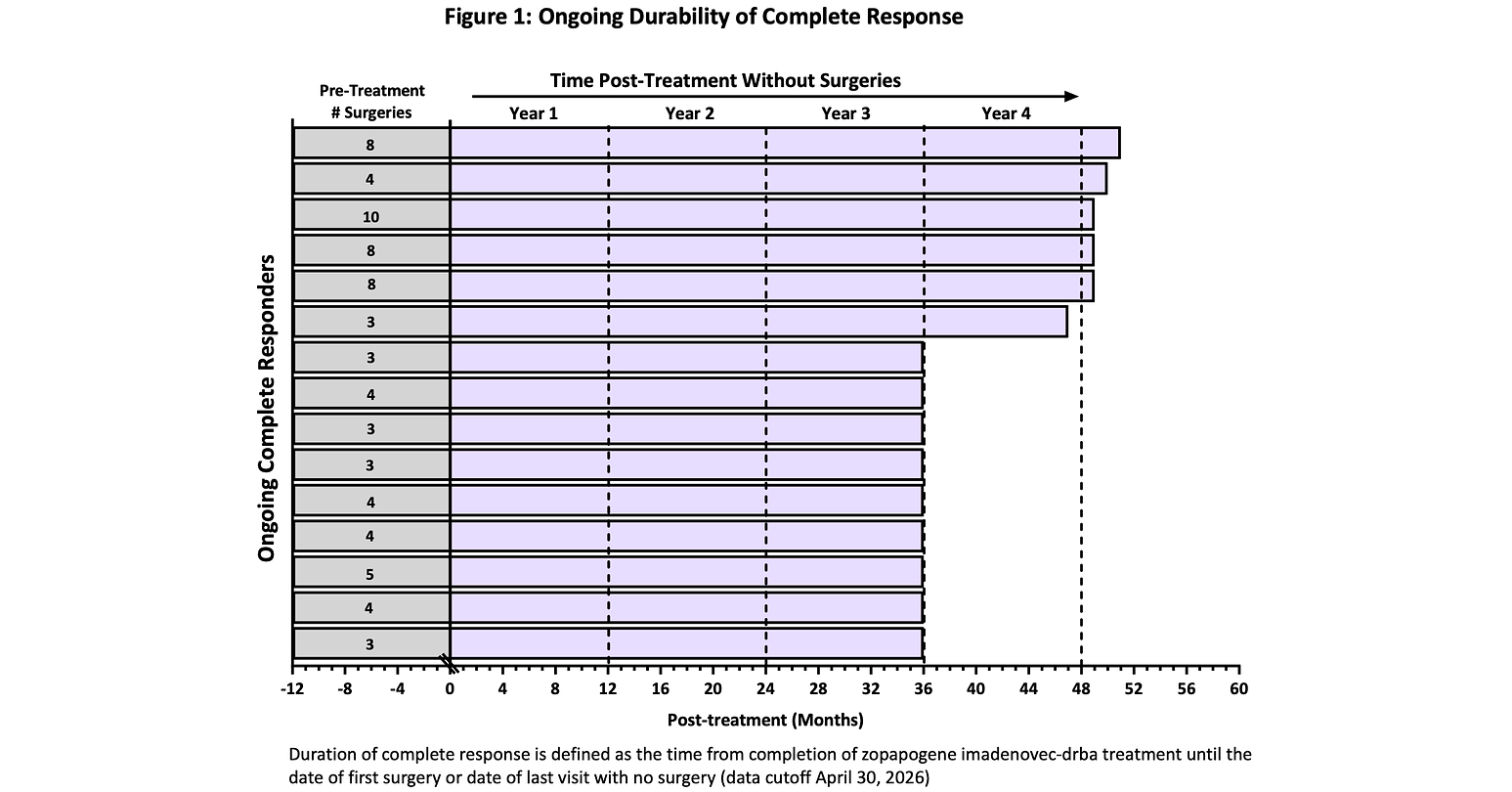
- Efficacy Durability: As of April 30, 2026, 15 out of 18 complete responders (83%) maintained ongoing complete responses without additional treatments, demonstrating the effectiveness and tolerability of PAPZIMEOS in treating recurrent respiratory papillomatosis (RRP).
- Long-Term Follow-Up Data: All complete responders had at least 36 months of follow-up, with an average of 40 months, and five patients have ongoing responses beyond four years, indicating the therapy's potential for long-term management of RRP.
- FDA Approval and Market Exclusivity: PAPZIMEOS received full FDA approval and was granted a seven-year orphan drug market exclusivity, further solidifying its position as the only approved therapy for RRP, which may drive future revenue growth for the company.
- Safety Assessment: No new adverse safety events were observed during long-term follow-up, supporting its role as a new standard of care and enhancing confidence in its safety profile.
- Durability of Efficacy: At the 2026 ASCO Annual Meeting, Precigen presented updated long-term follow-up data for PAPZIMEOS, revealing that all complete responders have been followed for over 36 months, with 83% maintaining response and five patients surgery-free for over four years, indicating the therapy's durability and potential to redefine treatment standards for RRP.
- Market Exclusivity: PAPZIMEOS has received full FDA approval and seven years of market exclusivity, establishing itself as the first and only approved therapy for adults with recurrent respiratory papillomatosis, thereby reinforcing Precigen's leadership position in the biopharmaceutical sector.
- Patient Impact Analysis: An internal analysis indicates approximately 27,000 adult RRP patients in the U.S., with traditional management relying heavily on repeated surgeries; the introduction of PAPZIMEOS not only alleviates the surgical burden on patients but also enhances quality of life and reduces healthcare system strain.
- Innovative Treatment Platform: PAPZIMEOS is developed using Precigen's proprietary AdenoVerse therapeutic platform as a non-replicating adenoviral vector-based immunotherapy designed to elicit immune responses against HPV 6 and HPV 11 proteins, representing a new paradigm in RRP treatment that may offer patients more effective long-term solutions.
- Orphan Drug Designation: The FDA has granted Precigen's PAPZIMEOS™ (zopapogene imadenovec-drba) orphan drug exclusivity, effective until August 14, 2032, ensuring seven years of market exclusivity in the US, thereby enhancing its competitive position and long-term revenue potential.
- First Approved Treatment: PAPZIMEOS received full FDA approval in August 2025, becoming the first and only drug approved for recurrent respiratory papillomatosis (RRP), addressing a significant treatment gap and expected to greatly improve patients' quality of life.
- Market Availability: PAPZIMEOS is now commercially available across the US, being prescribed in major medical centers and community practices, indicating its broad acceptance and potential for widespread clinical application among patients with varying disease severities.
- Innovative Treatment Paradigm: PAPZIMEOS utilizes a non-replicating adenoviral vector-based immunotherapy designed to generate an immune response against HPV 6 and HPV 11 proteins, representing a new therapeutic paradigm for RRP that could transform patient treatment approaches and outcomes.
- Investor Event Schedule: Precigen will participate in a J.P. Morgan virtual fireside chat on June 1, featuring CEO Dr. Helen Sabzevari in discussion with analyst Brian Cheng, aimed at enhancing the company's visibility in the biopharmaceutical sector.
- Global Healthcare Conference Participation: The company will also attend the Goldman Sachs 47th Annual Global Healthcare Conference from June 8 to 10 in Miami Beach, where Dr. Sabzevari will speak on June 8, showcasing the company's innovative drug pipeline.
- Innovative Drug Pipeline: Precigen focuses on developing precision medicines for difficult-to-treat diseases, committed to scientific breakthroughs from proof-of-concept to commercialization, which is expected to drive future revenue growth.
- Market Outlook: The company is actively advancing the commercialization of PAPZIMEOS™, aimed at treating adult recurrent respiratory papillomatosis, which, if successful, will significantly enhance Precigen's competitiveness in the biopharmaceutical market.
- Strong Financial Performance: Precigen reported $23.3 million in revenue for Q1 2026, a $21.9 million increase year-over-year, exceeding consensus estimates by $2.5 million, indicating robust demand for its newly launched cancer therapy, Papzimeos.
- Significant Reduction in Net Loss: The company's net loss fell approximately 85% year-over-year to $7.9 million, reflecting effective cost control and rapid revenue growth, which enhances investor confidence in its future profitability.
- Market Expansion Plans: CEO Helen Sabzevari stated that Precigen aims to explore opportunities for Papzimeos in overseas markets and the pediatric population, further driving revenue growth and increasing market share.
- Analyst Optimism: Seeking Alpha analyst Sean Daly reaffirmed his Buy rating on Precigen, noting that the company demonstrated real commercial validation in its Q1 earnings call, and he expects the market to recognize its future earnings potential.
- Price Target Increase: Citizens raised Precigen's price target from $9 to $11 while maintaining an 'Outperform' rating, indicating a potential upside of 165% from Wednesday's closing price, reflecting strong market confidence in the company.
- Sales Exceed Expectations: Precigen's Papzimeos generated $21.6 million in sales, significantly surpassing Wall Street's estimate of $20.8 million, contributing to a total revenue of $23.3 million in Q1, showcasing robust performance in the biotech sector.
- Investor Sentiment Soars: Precigen saw a surge of over 660% in message volume on Stocktwits in the past 24 hours, with investor sentiment shifting from 'bullish' to 'extremely bullish', indicating optimism surrounding its HPV cancer therapy candidate PRGN-2009.
- Positive Future Outlook: Precigen anticipates reaching cash flow break-even by the end of 2026, supported by existing cash reserves and revenue from Papzimeos sales, suggesting strong potential for sustainable growth in the coming years.