Cytokinetics Receives FDA Approval for MYQORZO, Driving Stock Surge
Cytokinetics Inc's stock surged by 8.64% as it reached a 5-day high, reflecting positive investor sentiment following recent developments.
The increase in stock price is attributed to Cytokinetics receiving FDA approval for MYQORZO (aficamten) to treat symptomatic obstructive hypertrophic cardiomyopathy (oHCM). This approval marks a significant milestone for the company, transitioning it from a development-stage biotech to a commercial entity. The approval is expected to enhance revenue prospects, addressing a critical gap in treatment options for oHCM patients.
This FDA approval not only boosts Cytokinetics' market position but also reflects the company's commitment to innovation in muscle biology. Investors are optimistic about the potential revenue growth as MYQORZO is anticipated to be available in the U.S. market soon.
Trade with 70% Backtested Accuracy
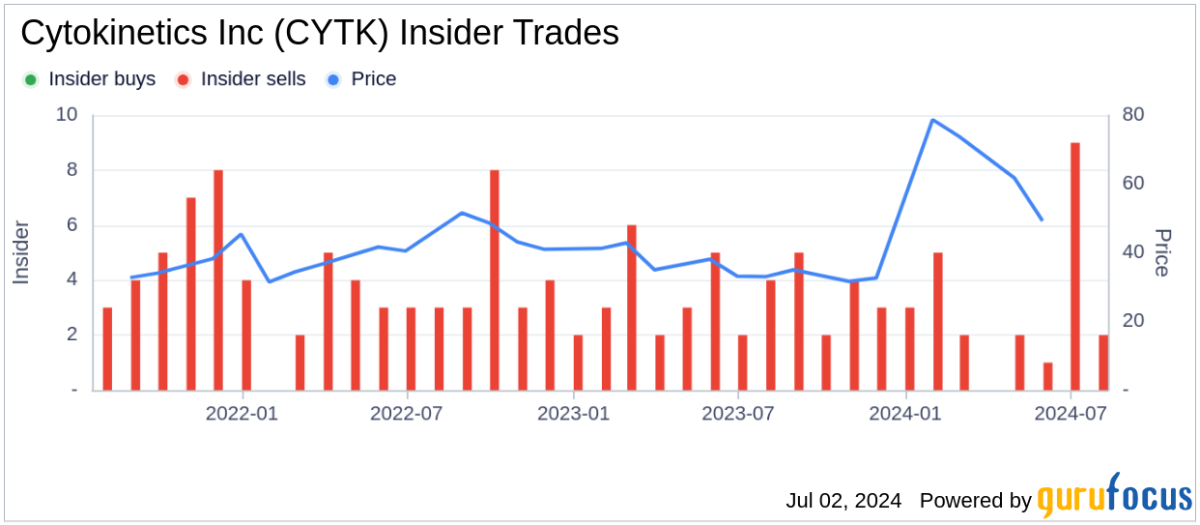
Analyst Views on CYTK
About CYTK
About the author

- Symposium Date and Venue: Cytokinetics will host the third annual Contemporary Landscapes in Muscle Biology Research Symposium on May 29, 2026, at the Mission Bay Conference Center in San Francisco, CA, from 8 AM to 5 PM Pacific Time, aimed at fostering dialogue and discovery in muscle biology.
- Registration Details: The symposium is free for attendees, but capacity is limited, requiring interested parties to register online by May 22, 2026, reflecting the company's commitment to advancing scientific communication.
- Keynote and Agenda: The event will feature keynote presentations from distinguished experts covering cardiac biology, skeletal muscle biology, and emerging treatment modalities, with the goal of driving advancements in the biological understanding and treatment of muscle-related diseases.
- Company Background and Development: Cytokinetics is a specialty cardiovascular biopharmaceutical company with over 25 years of pioneering innovations in muscle biology, currently developing multiple potential new medicines for cardiac muscle dysfunction, showcasing its ongoing investment and growth potential in the biopharmaceutical sector.
- Clinical Data Presentation: Cytokinetics showcased new clinical data for MYQORZO (aficamten) at the ESC Heart Failure 2026 Congress, emphasizing its effectiveness and safety compared to metoprolol, further solidifying its potential in treating symptomatic obstructive hypertrophic cardiomyopathy (oHCM).
- Sex Differences Analysis: The MAPLE-HCM trial revealed that MYQORZO demonstrated similar improvement effects in female patients compared to males, despite women entering the trial with more severe baseline characteristics, indicating the drug's consistent efficacy across genders, which may enhance personalized clinical treatment.
- Long-term Safety Data: A prospective analysis from FOREST-HCM indicated that long-term treatment with MYQORZO for up to 96 weeks did not increase the incidence of arrhythmias, reinforcing its safety profile, particularly in patients who discontinued beta-blocker therapy, thereby boosting confidence in its clinical application.
- Left Atrial Remodeling Improvement: Expanded insights from SEQUOIA-HCM provided the first analysis of MYQORZO's effects on left atrial function, showing significant improvements in left atrial mechanics, potentially offering new therapeutic avenues for enhancing functional capacity in oHCM patients.
- Offering Size: Cytokinetics successfully closed a public offering of 11,338,028 shares at $71.00 per share, raising approximately $805 million, indicating strong market demand for its stock.
- Underwriter Lineup: The offering was managed by Morgan Stanley, Goldman Sachs, J.P. Morgan, and Jefferies, reflecting confidence from investment banks in Cytokinetics' future growth and potentially enhancing its market position.
- Registration Statement: The securities were offered under a shelf registration statement filed with the SEC on February 27, 2025, ensuring compliance and streamlining the offering process, which boosts investor confidence.
- Business Context: Cytokinetics focuses on cardiovascular biopharmaceuticals, developing potential new medicines for cardiac muscle dysfunction, and the raised funds will support its R&D pipeline, particularly innovative treatments for heart failure.
- Upsized Offering: Cytokinetics successfully priced an upsized public offering of 9.86 million shares at $71 per share, raising approximately $700 million in gross proceeds before expenses, indicating strong market demand for its stock.
- Underwriter Option: The underwriters were granted a 30-day option to purchase up to an additional 1.48 million shares, further enhancing market confidence in Cytokinetics' future growth potential.
- Closing Timeline: The offering is expected to close on May 8, marking an active financing strategy for the company in the capital markets aimed at supporting its R&D and market expansion plans.
- Operating Expense Outlook: Earlier this week, Cytokinetics announced a proposed $650 million stock offering and outlined an operating expense range of $830 million to $870 million for 2026, reflecting the company's strategic planning for future growth.
- Offering Size: Cytokinetics announced a public offering of 9,859,155 shares at $71 per share, aiming for gross proceeds of approximately $700 million, which will significantly bolster the company's funding for research and operations.
- Underwriter Lineup: Morgan Stanley, Goldman Sachs, J.P. Morgan, and Jefferies are acting as joint book-running managers for the offering, reflecting strong market confidence in Cytokinetics and enhancing the company's reputation in capital markets.
- Additional Share Option: Cytokinetics has granted underwriters a 30-day option to purchase an additional 1,478,873 shares at the public offering price, providing flexibility that may further enhance the company's liquidity.
- Market Outlook: The offering is expected to close on May 8, 2026, and if successful, will provide robust financial support for Cytokinetics' cardiovascular biopharmaceutical research, aiding in the development of innovative drugs for cardiac muscle diseases.
- Company Update: Cytokinetics has raised its target price to $146.00 from $108.00.
- Market Reaction: This adjustment reflects positive market sentiment and expectations regarding the company's performance.





